Regulatory Oversight Report on the Use of Nuclear Substances in Canada: 2016
Regulatory Oversight Report on the Use of Nuclear Substances in Canada: 2016 (PDF 4475 kb)
Table of Contents
- Executive Summary
- 1 Background
- 2 Report overview
- 3 Regulatory program for the use of nuclear substances
- 3.1 CNSC regulatory effort
- 3.2 Licensing
- 3.3 Certification of prescribed equipment
- 3.4 Certification of exposure device operators
- 3.5 Certification of Class II radiation safety officers
- 3.6 RSO appointment for nuclear substances and radiation device licences
- 3.7 Licensing and certification decisions
- 3.8 Compliance verification and enforcement
- 3.9 Stakeholder engagement
- 4 Regulatory developments
- 5 Safety performance – all sectors combined
- 6 Medical sector
- 7 Industrial sector
- 8 Academic and research sector
- 9 Commercial sector
- 10 Conclusion
- Appendix A: Radiation exposure
- Appendix B: Safety and control area naming conventions
- Appendix C: Enforcement actions issued in 2016
- Appendix D: List of reported events in 2016
- Appendix E: Inspections conducted in 2016
- Appendix F: Compliance rating levels
- Appendix G: Grading inspections
- Appendix H: Abbreviations and glossary
- Appendix I: Relevant regulatory references
Executive summary
The Regulatory Oversight Report on the Use of Nuclear Substances in Canada: 2016 summarizes the safety performance of 1,584 licensees that hold a total of 2,233 licences, and which are authorized by the Canadian Nuclear Safety Commission (CNSC) for the use of nuclear substances in the medical, industrial, academic and research, and commercial sectors.
The CNSC regulates the nuclear industry in Canada through a comprehensive program of licensing, certification, compliance verification, enforcement and reporting. For each sector described in this report, CNSC staff evaluate safety performance through inspections, assessments, and reviews of licensee programs and processes.
CNSC staff use a well-established safety and control area (SCA) framework in evaluating each licensee’s safety performance. The framework includes 14 SCAs covering all technical areas of regulatory oversight. For the purpose of this report, safety performance is demonstrated by examining licensees’ regulatory compliance in select SCAs (i.e., management system, operating performance, radiation protection, and security), as well as effective doses to workers and reported events.
In 2016, as part of the ongoing regulatory oversight of licensees, CNSC staff conducted compliance verification activities consisting of field inspections, desktop reviews and technical assessments of licensee activities. The evaluations of findings for the SCAs covered in this report show that, overall, licensees made adequate provisions for the protection of the health, safety and security of persons and the environment from the use of nuclear substances, and took the measures required to implement Canada’s international obligations. Based on this, CNSC staff conclude that the use of nuclear substances in Canada is safe.
Compliance verification
In 2016, CNSC staff conducted 1,452 inspections across the four sectors, including 228 security inspections related to the implementation of REGDOC-2.12.3, Security of Nuclear Substances: Sealed Sources, and three inspections related to the export of high-risk sealed sources.
Overall, licensees showed satisfactory compliance ratings in all SCAs examined for this report. CNSC staff ensured that licensees took appropriate corrective actions in cases where non-compliances were discovered. Any non-compliance that had immediate risks to health, safety or security were addressed immediately by licensees. The majority of inspected licensees in 2016 were found to be compliant with requirements in the four SCAs covered in this report:
- In management system, 97.5 percent of licensees ensured that adequate processes and programs were in place to achieve their safety objectives.
- In operating performance, 87.4 percent of licensees made adequate provisions for the health, safety and security of persons, and protection of the environment.
- In radiation protection, 84.6 percent of licensees ensured that exposure of workers and the public to ionizing radiation remained as low as reasonably achievable.
- In security, 93.6 percent of licensees demonstrated that they have adequate provisions in place to prevent the loss, sabotage, illegal use, illegal possession or illegal removal of nuclear substances and prescribed equipment in their care and control.
As part of the phased implementation of REGDOC-2.12.3, security inspections were conducted for licensees in possession of high-risk sources in 2016. Of those inspected, 79 percent were found to be compliant with the regulatory requirements. Licensees have put in place measures to correct all non-compliances noted during these inspections. Details of the security inspections, such as non-compliances, are not provided in this report due to their sensitive nature.
Compliance enforcement
The CNSC uses a graded approach to enforcement to encourage and compel compliance and deter future non-compliances. When non-compliance (or continued non-compliance) has been identified, CNSC staff assess the significance of the non-compliance, and determine the appropriate enforcement action, based on the CNSC’s graded approach to enforcement.
In 2016, the CNSC took 22 escalated compliance enforcement actions against licensees in the four sectors, including 14 orders and eight administrative monetary penalties. Most of the enforcement actions were taken against licensees in the industrial sector, consistent with trends from previous years. CNSC staff reviewed corrective measures implemented by all licensees to whom orders were issued and found them to be satisfactory. All eight administrative monetary penalties issued in 2016 have been paid.
Effective doses to workers
Licensees are required to keep radiation doses to persons below CNSC regulatory limits and as low as reasonably achievable in accordance with the radiation protection programs established under the CNSC licences.
In 2016, doses were monitored for 62,013 workers in the four sectors covered in this report. Of those workers, 22,606 were designated as nuclear energy workers (NEWs). The remaining 39,407 were not designated as NEWs, and are referred to as non-NEWs in the report. Exposures to radiation continued to be very low for workers in 2016, consistent with previous reporting years.
As reported to the Commission in December 2016, one NEW received an extremity dose of 1,100 millisieverts (mSv) from contamination while administering medical isotopes. That dose was above the regulatory limit of 500 mSv. Additional details can be found in section 5.7 of this report. None of the workers designated as NEWs exceeded the one-year effective dose limit of 50 mSv. Also as reported to the Commission in December 2016, one member of the public received a dose of 1.62 mSv as a result of the transport of packages containing nuclear substances with passengers. That dose exceeded the regulatory limit of 1 mSv.
Reported events
For 2016, CNSC staff assessed the 139 events reported by licensees covered in this report. Reported events have been ranked using the International Nuclear and Radiological Event Scale. Of these, 136 were ranked as level 0 (no safety significance), two were ranked as level 1 (anomaly) and one was ranked as level 2 (incident).
For all of the events reported, licensees implemented appropriate response measures to mitigate the impacts of the events and to limit radiation exposure to workers and the public. CNSC staff reviewed the measures and found them to be satisfactory.
Conclusion
As a result of the CNSC’s comprehensive regulatory oversight of the industry, CNSC staff conclude that the use of nuclear substances in Canada is safe. Adequate provisions for the protection of the health, safety and security of persons and the environment from the use of nuclear substances are in place.
1 Background
1.1 Background
The Canadian Nuclear Safety Commission (CNSC) regulates the use of nuclear energy and materials to protect health, safety, security and the environment; to implement Canada’s international commitments on the peaceful use of nuclear energy; and to disseminate objective scientific, technical and regulatory information to the public. Persons licensed by the CNSC are responsible for operating their facilities and managing their activities safely and are required to implement programs that make adequate provisions for protecting health, safety, security and the environment. The CNSC is responsible for setting the requirements and verifying compliance against those requirements.
Each year, CNSC staff assess the overall safety performance of the use of nuclear substances in Canada. Staff consider industry performance as a whole, as well as the performance of each sector (i.e., medical, industrial, academic and research, and commercial) separately. This assessment is summarized in this document.
For a comprehensive overview of the CNSC and its activities, consult the CNSC’s annual report, Regulating Nuclear Safety in Canada.
Regulatory oversight
The CNSC regulates the nuclear industry in Canada through a comprehensive program of licensing, certification, compliance verification, and enforcement. For each of the nuclear sectors described in this report, CNSC staff evaluate safety performance through assessments, inspections, reviews and evaluations of licensee programs and processes.
These regulatory programs cover various types of activities across all provinces and territories, as shown in figure 1. Licensees include hospitals, universities and research institutions, and a wide variety of industrial manufacturing and production facilities including those that store, produce or service nuclear substances and devices.
The safe use of nuclear substances in Canada is demonstrated through licensees’ compliance with the Nuclear Safety and Control Act (NSCA) as well as its associated regulations, and specific conditions set out in CNSC licences. The NSCA, its regulations and the licences require that licensees implement and maintain appropriate programs to ensure the safety and security of nuclear-related activities, minimize doses to workers and the public, protect the environment, and minimize consequences of events.
.jpg/object)
Some licensees that hold CNSC licences to service radiation devices or prescribed equipment are based outside Canada and come to Canada to perform maintenance or servicing work on equipment owned by other licensees. When they work in Canada, these licensees are subject to the same level of regulatory oversight from the CNSC as those whose operations are based in Canada.
Safety and control area framework
To ensure comprehensive regulatory oversight and reporting of licensed activities, CNSC staff have developed a set of safety and control areas (SCAs). SCAs have been in use for a number of years, and represent a well-established set of technical areas that have proven effective in evaluating licensee safety performance of regulated facilities and activities under the CNSC’s purview. The CNSC has defined 14 SCAs:
- management system
- human performance management
- operating performance
- safety analysis
- physical design
- fitness for service
- radiation protection
- conventional health and safety
- environmental protection
- emergency management and fire protection
- waste management
- security
- safeguards and non-proliferation
- packaging and transport
2 Report overview
This regulatory oversight report focuses on the results of compliance verification and enforcement activities in 2016 for licensees that use nuclear substances in four sectors:
- medical
- industrial
- academic and research
- commercial
Each sector performance is outlined in an individual section in this report.
The report does not cover uranium mines and mills, waste facilities, dosimetry services, Class I nuclear facilities such as nuclear power plants and nuclear research reactors, or Class IB accelerator facilities.
There are three parts to this report:
- regulatory process and developments
- overall safety performance assessment
- sector-specific safety performance assessments
2.1 Safety performance measures
CNSC staff review licensee documents and conduct field inspections to verify that licensees have implemented effective safety programs and practices. Results of these inspections provide information on key aspects of safety performance, within each SCA relevant to the licensed activity.
For the purpose of this report, the following four SCAs are the most relevant indicators of safety performance for licensees in the sectors covered in this report: management system, operating performance, radiation protection, and security. Compliance ratings – also referred to as inspection ratings – reflect overall licensee performance at a program level for each of these SCAs. The nature, type and safety significance of events reported by licensees, and the type of enforcement actions taken by the CNSC in 2016 are provided as supplementary indicators of safety performance. Data from 2012 to 2016 are included in figures for each of these safety indicators to identify five-year trends. Each performance measure is described below.
Appendix B shows the mapping between the CNSC regulatory naming convention in the inspection reports and those presented in this report for SCAs.
2.1.1 Doses to workers
Each licensee is required to implement a radiation protection program that ensures that the radiation doses to workers are well below regulatory limits and kept as low as reasonably achievable (ALARA), with social and economic factors taken into account. Thus, ascertainment of the magnitude of doses received by workers is an integral part of a licensee’s radiation protection program.
This report references two groups of workers that perform the types of work referenced in a CNSC licence: those designated as nuclear energy workers (NEWs) and those that are not designated as NEWs (non-NEWs). The term NEW means a person who is required, in the course of his or her business or occupation in connection with a nuclear substance or nuclear facility, to perform duties in circumstances that may result in receiving a dose of radiation greater than 1 millisievert (mSv) per year. A worker not designated as a NEW means a person is unlikely to receive a dose greater than 1 mSv per year while performing duties in connection with a nuclear substance or nuclear facility. This report provides dose information for all workers, while primarily focusing on those designated as NEWs.
The CNSC’s regulatory effective dose limits for NEWs are set at 50 mSv in any one-year dosimetry period and a total of 100 mSv over a five-year dosimetry period. The one-year dosimetry period covers January 1 to December 31 of every year. The current five-year dosimetry period started on January 1, 2016 and will end on December 31, 2020. For all persons not designated as NEWs, and for all members of the general public, the effective dose limit is 1 mSv per calendar year.
For activities where there is a need for direct handling of nuclear substances, doses to the hands are also monitored. These are known as extremity doses, and they are subject to a regulatory dose limit of 500 mSv in any one-year dosimetry period for NEWs and 50 mSv per calendar year for workers not designated as NEWs. The concept of a five-year dosimetry period does not apply to extremity doses or effective doses incurred by persons who are not NEWs.
Appendix A provides more information on occupational exposure, ascertaining worker doses and measures to be taken by licensees when a dose limit is exceeded.
2.1.2 Management system
The management system SCA covers the framework that establishes the processes, programs and resources required to ensure that a licensee achieves its safety objectives, continuously monitors its performance against those objectives, and fosters a healthy safety culture.
2.1.3 Operating performance
Operating performance refers to the licensee’s ability to perform licensed activities in accordance with pertinent operational and safety requirements defined in the NSCA, its associated regulations and licence conditions. Licensees are expected to demonstrate that they comply with operational and safety requirements by providing workers with appropriate procedures for the safe use of nuclear substances and prescribed equipment, by ensuring that workers follow procedures, and by maintaining records that demonstrate compliance.
2.1.4 Radiation protection
Radiation protection programs are required for every licensee to ensure that contamination levels and radiation doses received by workers are monitored, controlled and maintained below regulatory dose limits, and kept ALARA, with social and economic factors taken into account. Licensees can meet these objectives by monitoring worker doses; posting radiation warning signs; planning appropriately for radiological emergencies; managing oversight of operational activities; instituting effective workplace practices that emphasize the use of time, distance and shielding to minimize exposure to radiation; and using appropriate protective equipment.
2.1.5 Security
The security SCA covers the physical security measures, practices and programs that licensees are required to have in place to prevent the loss, illegal use, illegal possession or illegal removal of nuclear substances during their entire lifecycle, including while they are in storage or during transport. The extent of the security measures required depends upon the types of nuclear substances used and activities performed by each licensee.
The safety and security of sealed sources is increased through effective control and tracking. CNSC compliance inspections include requirements to verify sealed source tracking information.
To ensure proper regulatory oversight of the requirements related to the phased implementation of REGDOC-2.12.3, Security of Nuclear Substances: Sealed Sources, CNSC staff conduct security inspections for those in possession of Category 1 and 2Footnote 1 sealed sources. Non-compliance details for these inspections are not included in this report due to their sensitive nature.
2.1.6 Enforcement actions
The CNSC may take a variety of enforcement actions to ensure that licensees correct non-compliances in an effective and timely manner. The type of enforcement action taken is commensurate with the risk the non-compliance presents to the environment, the health and safety of workers and the public, and to national security. This report provides detailed information on the following types of enforcement actions taken by the CNSC: orders, administrative monetary penalties (AMPs), decertification of certified exposure device operators and decertification of radiation safety officers at Class II nuclear facilities. Appendix C provides a list of all orders and AMPs issued by the CNSC in 2016. No exposure device operators or radiation safety officers were decertified in 2016.
2.1.7 Reported events
Under the NSCA and its associated regulations, licensees are required to immediately report to the CNSC events related to their licensed activities that are of regulatory interest. Within 21 days of becoming aware of the incident, licensees are required to submit a final report to the CNSC on the event. The final report must include an analysis of the cause and circumstances of the event, as well as any measures taken, or proposed to be taken, by the licensee to prevent recurrence. Together, the initial and final reports allow the CNSC to verify whether the licensee has taken appropriate measures to mitigate the event, and implemented adequate corrective actions to prevent recurrence.
The CNSC uses the International Nuclear and Radiological Event Scale (INES) tool to categorize events in the sectors covered by this report. Additional information on the INES classification can be found on the CNSC website.
2.2 Data collection
Compliance ratings, non-compliance data, and CNSC enforcement actions were obtained from the CNSC’s compliance verification and enforcement program in 2016.
Annual compliance reports submitted by licensees in calendar year 2016 provided the data on doses incurred by all persons engaged in licensed activities in the four sectors covered in this report.
2.3 Changes in 2016
At the Commission’s direction, CNSC staff introduced the following additions to this report from those of previous years:
- The frequency of repeated unacceptable SCA ratings for licensees is included in the report.
- A list of all inspections conducted in 2016 is included in appendix E.
- Additionally, information on the number of international events involving nuclear substances and radiation devices reported to the International Atomic Energy Agency has been included in the report.
3 Regulatory program for the use of nuclear substances
The possession, use, transfer, import, export, abandonment and storage of nuclear substances must be licensed by the CNSC when the amount of nuclear substance involved is greater than its exemption quantity (see Schedule 1 of the Nuclear Substances and Radiation Devices Regulations). Facilities where certain types of Class II prescribed equipment are to be installed must also be licensed by the CNSC prior to their construction, operation or decommissioning. A licence is also required to service radiation devices or Class II prescribed equipment.
All licensees that operate Class II nuclear facilities or that service Class II prescribed equipment must have a certified radiation safety officer and a qualified temporary replacement. The radiation safety officer has the responsibility to implement the radiation protection program, ensuring that licensed activities are conducted safely and that all regulatory requirements are met.
All radiation devices and most Class II prescribed equipment, as well as certain types of transport packages, must be certified by the CNSC before they can be used in Canada.
An integral part of the CNSC’s regulatory oversight is the compliance and verification program, which measures licensee compliance with CNSC regulatory requirements. Regular inspections and desktop evaluations verify that licensees comply with the Nuclear Safety and Control Act (NSCA) and its associated regulations, as well as any conditions included in their licences.
To determine appropriate levels of regulatory monitoring and control, CNSC staff establish compliance verification plans for each nuclear sector that are based on risk-informed regulatory oversight of each sector’s activities. Modifications to the compliance plans are made on an ongoing basis in response to events and changes in licensees’ performance.
For the activities covered in this report, the CNSC’s risk-informed regulatory program is applied in the following way:
- Each licensed activity is assigned a weighting factor – a coefficient that represents the activity’s relative significance with respect to risk.
- Factors considered in weighting include the form of the nuclear substances (e.g., sealed source, unsealed source or radiation device), the location where the material is being used (e.g., a work site or a controlled facility), and the compliance history of licensees conducting licensed activities.
- Generally, licensees are inspected on a one to five year cycle, based on their risk ranking.
The risk-informed regulatory program provides:
- a risk ranking that recognizes the potential safety impact of the licensed activity
- effective and informed allocation of regulatory oversight effort according to the risk ranking by licensed activity and by licensee performance history
- effective, transparent, consistent and comprehensive regulatory oversight
3.1 CNSC regulatory effort
The CNSC’s risk-informed regulatory program applies resources and regulatory oversight commensurate with the risk associated with the regulated activity. Regulatory effort related to licensing, certification and compliance verification is derived from this program. A total of 1,452 inspections were completed in 2016. As shown in table 1, the CNSC staff direct effort for regulating the use of nuclear substances in 2016 amounted to close to 12,645 person days or the annual equivalent of approximately 56 full-time staff. This is down slightly from the staff effort in 2015 which amounted to 59 full-time staff.
| Activity | Person days |
|---|---|
| Licensing | 4,451 |
| Certification | 1,790 |
| Compliance verification | 6,404 |
3.2 Licensing
To obtain a licence, an applicant must submit an application to the CNSC. The CNSC will issue a licence only when the applicant:
- is deemed qualified to carry on the activity that the licence will authorize
- has demonstrated that it will protect the health and safety of persons and the environment
- has demonstrated that it will maintain national security
- has confirmed that it will adhere to international obligations to which Canada has agreed
CNSC staff perform a risk-informed technical assessment of applications submitted to the CNSC.
The CNSC has produced a series of licence application guides to ensure that its expectations for applicants are clear, and to facilitate applicants’ interactions with the regulator. These guides are reviewed on a five-year cycle as part of the regulatory framework review to ensure they continue to reflect modern regulatory expectations and provide useful guidance to the regulated community. This practice, in turn, facilitates CNSC licensing reviews and minimizes regulatory burden. Application forms and guides can be found on the CNSC website for nuclear substances and radiation devices as well as for Class II facilities and prescribed equipment.
When applying for licence renewals, existing licensees are subject to the same scrutiny as new applicants. The CNSC decision to renew a licence is based on the application information submitted as well as a satisfactory compliance history. This includes a review of compliance information such as inspection results, reported incidents and events, and annual compliance reports.
If the application satisfies the above requirements, the Commission, or a designated officer authorized by the Commission, may issue a licence authorizing the licensee to conduct the activities requested in the application. The licence includes provisions that define and limit the scope of the authorized activities, as well as specific conditions that must be fulfilled by the licensee when conducting those activities.
3.2.1 Licence consolidation strategy
The Directorate of Nuclear Substance Regulation (DNSR), in line with the CNSC’s policy to consolidate licences, has developed a strategy aimed at reducing administrative burden on organizations that hold multiple licences for various licenced activities such as hospitals and universities. This strategy included creating and issuing new consolidated licences for Class II nuclear facilities as well as reviewing the licence format and process for those that use nuclear substances and radiation devices.
Consolidation of Class II nuclear facility licences has allowed the CNSC to, for example, authorize a hospital with a medical linear accelerator to operate and service the accelerator under one licence instead of two. In other cases, it has allowed cancer centres to include several types of radiotherapy activities to be covered under a single licence instead of as many as five. In some cases, such as a single organization operating both cyclotron and radiation therapy facilities, licence consolidation is not desired since each licensee’s radiation protection program and management structure is different.
The majority of licences issued through the Directorate of Nuclear Substance Regulation are for the possession and use of nuclear substances and radiation devices. A comprehensive evaluation of the licensing process was conducted in 2016 as the first part of the implementation strategy to streamline the application process and reduce the number of licences required. The revised licensing process will be implemented in 2017. It will be accompanied by an updated version of REGDOC-1.6.1, Licence Application Guide: Nuclear Substances and Radiation Devices, which was published in May 2017 and which provides greater clarity and simplified requirements for applicants. Due to the number of licences and the volume of ongoing work, changes to the licensing approach must be carefully considered to minimize the impact on licensees.
The number of licences issued by sector is shown in table 2 and the distribution of licences by select groups is provided in figure 2. Most licensees, as represented by the industrial sector, perform one licensed activity and, therefore, require only one CNSC licence. Hospitals tend to conduct various licensed activities, such as diagnostic nuclear medicine, cancer therapy, production and processing of nuclear medicine isotopes and human research. These disparate activities are covered under specific CNSC licences which reflect the unique characteristics of such uses. As a result, hospitals typically hold more than one CNSC licence. Moreover, a number of provinces are in the process of amalgamating the administration of several hospitals under new authorities. CNSC staff work with these licensees to ensure that there is the appropriate level of regulatory control while minimizing administrative burden wherever possible, meaning the workload for CNSC staff has not changed significantly as a result of licence consolidation.
As stated in the previous paragraph, the workload of the CNSC staff remained the same while the number of licences issued by the DNSR continues to decrease. This is driven by a combination of licence consolidation strategies put in place at the CNSC and changes in the industry, including mergers and acquisitions.
| Sector | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Medical | 561 | 552 | 536 | 494 | 470 |
| Industrial | 1,451 | 1,440 | 1,398 | 1,349 | 1,308 |
| Academic and research | 253 | 232 | 229 | 207 | 208 |
| Commercial | 248 | 256 | 248 | 245 | 247 |
| Total | 2,513 | 2,480 | 2,411 | 2,295 | 2,233 |
.jpg/object)
Description
| Sector | 1 | 2-3 | 4-6 | 7-9 | 10+ |
|---|---|---|---|---|---|
| Hospitals | 26 | 78 | 31 | 5 | 4 |
| Industrial sector | 974 | 111 | 12 | 5 | 0 |
| All sectors combined | 1252 | 262 | 54 | 12 | 4 |
3.3 Certification of prescribed equipment
An application for certification must be submitted to the CNSC before the prescribed equipment can be used in Canada. CNSC staff who conduct the technical evaluations of applications for certification are accredited as professional engineers as part of their job requirements. Upon receipt of an application, CNSC staff conduct a thorough technical review of the information contained in the submission to determine if:
- the radiation device, Class II prescribed equipment or transport package meets all CNSC regulatory requirements and is safe to use
- adequate measures are in place in respect of their use to protect the environment, national security, and the health, safety and security of persons
A CNSC quality assurance program, in the form of a peer review by another CNSC staff member, is in place for the review of new applications for certification of prescribed equipment and those where significant changes are made to the original design.
If satisfied that the design meets the above requirements, the Commission, or a designated officer authorized by the Commission, may issue a certificate for the radiation device, Class II prescribed equipment or transport package based on the recommendation of the CNSC staff members who conducted the technical evaluations.
If the design does not comply with the above requirements or if a certified model is found to be unsafe, the designated officer will contact the applicant and all affected parties, such as users in the case of a certified model, to inform them of the decision to either not certify the new model or to decertify a currently certified model. In these cases, the CNSC will provide the applicant and affected parties an opportunity to be heard in accordance with the process specified in the regulations.
Regulatory documents REGDOC-1.5.1, Application Guide: Certification of Radiation Devices or Class II Prescribed Equipment (under revision, previously RD/GD-254) and RD/GD-352, Design, Testing and Performance of Exposure Devices outline CNSC expectations for the certification of radiation devices and Class II prescribed equipment, while RD/GD 364, Joint Canada – United States Guide for Approval of Type B(U) and Fissile Material Transportation Packages (currently under review), outlines CNSC expectations for the certification of transport packages.
3.4 Certification of exposure device operators
Licensees are required under the Nuclear Substances and Radiation Devices Regulations to permit only CNSC-certified personnel and supervised trainees to use exposure devices containing nuclear substances. In 2016, the CNSC certified 115 new exposure device operators (EDOs) and renewed the certifications of 340 others. EDOs must renew their certification every five years to ensure they maintain the knowledge and skills required to operate an exposure device safely.
The CNSC EDO-certification program is designed to ensure the continued competency of the operator, and maintain the safety and security of persons and devices when working with exposure devices. Certified individuals must demonstrate the ability to:
- handle, transport, store and operate exposure devices and any accessories to the devices safely and securely
- properly utilize radiation detection and monitoring equipment
- understand the obligation to comply with all relevant regulatory requirements
Regulatory document REGDOC-2.2.3, Personnel Certification: Exposure Device Operators and the CSA Group document CSA PCP-09 Certified Exposure Device Operator Personnel Certification Guide outline the CNSC’s requirements and guidance for certification as an EDO and for renewal of an EDO certification.
The CNSC may take regulatory action if an EDO is found to be operating contrary to safety protocols and conditions, or if an EDO is causing undue risk to the public or the environment. No EDOs were decertified in 2016.
3.5 Certification of Class II radiation safety officers
All licensees that operate Class II nuclear facilities or that service Class II prescribed equipment must have a certified radiation safety officer (RSO) and a qualified temporary replacement. The RSO ensures that licensed activities are conducted safely and all regulatory expectations are met.
There are two components to the RSO certification process:
- an assessment of the candidate’s capabilities to perform the duties of the position, based on the submitted application
- an assessment of the candidate’s knowledge of the licensed activities, based on an examination
RSO candidates must possess certain qualifications before they can be considered for certification. For most Class II licensed activities, candidates must have at least a bachelor’s degree in engineering or science from a recognized university.Footnote 2 Alternative education qualifications may be reviewed on a case-by-case basis.
If the candidate is able to clearly demonstrate their knowledge as it relates to the RSO position within their organization, the Commission or a designated officer authorized by the Commission may certify the candidate in the position of RSO.
The process for certification of Class II RSOs, along with guidance for applicants, is outlined in REGDOC-2.2.3, Personnel Certification: Radiation Protection Officers. In 2016, the CNSC certified 22 applicants as Class II RSOs. No Class II RSOs were decertified in 2016. The CNSC has certified 212 Class II RSOs since 2010.
3.5.1 Class II RSO examination
In 2016, the CNSC used an online examination for the first time to certify Class II RSOs.
The content of the examination focuses on five subjects:
- relevant provisions of the NSCA and its ensuing regulations
- principles of radiation safety
- radiation physics
- operational activities and facilities which are to be licensed by the CNSC
- radiation protection program of the facility
The content of the examination is tailored to:
- the operational risks of the licensed activity
- the organization’s policies and procedures
- the candidate’s academic background and work experience
3.6 RSO appointment for nuclear substances and radiation device licences
There are approximately 1,860 RSOs appointed for nuclear substances and radiation device licences. The designation of an RSO for nuclear substances and radiation devices licences is the responsibility of the applicant authority, the person accountable for the management and control of the licensed activity. The RSO is the person the CNSC will contact about radiation safety and compliance matters. The appointment of these RSOs does not involve a certification process.
The CNSC requires the RSO’s qualifications be included in a licence application and will determine if the RSO has sufficient knowledge and expertise with regard to the applicant’s proposed activities. The RSO may be a consultant hired by the applicant to carry out this role, provided that the consultant is clearly designated by the applicant authority to do so. Such information must be communicated to the CNSC as part of the licence application process. Alternate RSOs may be utilized where a licensee has multiple locations of licensed activity.
Unless otherwise noted by the applicant authority, the RSO will be considered to have the authority to act for the applicant and will have signing authority for all matters encompassed by the CNSC licence.
In 2017, CNSC staff started reviewing the oversight process of RSOs that are appointed (i.e., those that do not need to sit and pass an examination) to identify factors that may lead to greater success in that position. This process will be undertaken using internationally accepted methods and will be designed with the assistance of experts internal and external to the CNSC.
In addition to the evaluation project, the CNSC is producing a document that will provide greater guidance to those people performing in the role of RSO as to the CNSC expectations for individuals occupying the position of RSO. Publication of this document is tentatively scheduled for the end of 2018.
For high-risk activities, CNSC staff perform additional verifications. They meet with the applicant RSO and the applicant authority during a pre-licensing visit to verify the RSO’s knowledge of the company’s radiation protection program and confirm the applicant understands their obligations as a licensee. Staff plan visits and prepare the interview following review of the application and the applicant’s radiation protection program. During the visit, CNSC staff ensure the licensee understanding of the radiation safety program that has been committed to and review the location of the proposed licensed activities while ensuring that candidates have strong radiation safety knowledge as well as advanced training in operational and emergency procedures. If it is deemed that the appointed RSO does not have adequate knowledge, the licensing decision will be pending on the appointment of a suitable RSO. In 2016, CNSC staff performed eight pre-licensing visits for prospective nuclear substance and radiation device licensees.
3.7 Licensing and certification decisions
CNSC designated officers made a total of 2,805 licensing and certification decisions related to activities covered in this report in 2016. The majority of these were licensing decisions, as shown in Table 3.
There was a significant increase in the number of certification activities in 2016 due to the expiry and renewal of a higher-than-average number of device certificates. This trend will continue into 2017. In addition, there was an increase in the number of EDO certification decisions as existing EDOs sought renewals as per CSA PCP-09 – Certified Exposure Device Operator Personnel Certification Guide.
| Type of decision | Number of decisions |
|---|---|
| Licensing (issuance of new licences, licence renewals, licence amendments, licence revocations and licence transfers) | 2,185 |
| Certification of prescribed equipment (radiation devices, Class II prescribed equipment and transport packages) | 143 |
| Certification of EDOs (issuance of new certification and renewal of certification) | 455 |
| Certification of Class II RSOs | 22 |
| Total | 2,805 |
3.8 Compliance verification and enforcement
The CNSC verifies compliance by conducting site inspections and reviewing licensee documentation and operational activities. Licensees are required to report routine performance data through annual compliance reports and the occurrence of specific types of events. In addition, the CNSC conducts investigations of unplanned events, public complaints or accidents involving nuclear substances.
The CNSC uses a graded approach to enforcement to encourage compliance and deter future non-compliances. When a non-compliance (or a continued non-compliance) has been identified, CNSC staff assess its risk and safety significance to determine appropriate enforcement action. The chosen enforcement action is commensurate with the risk that the non-compliance presents to the environment, the health and safety of workers and members of the public, and to national security. Enforcement actions vary with non-compliance severity, and can include orders and administrative monetary penalties. Each is a discrete and independent response to a non-compliance.

Escalated enforcement actions were taken against licensees in the medical, industrial, academic and research, and commercial sectors in 22 instances in 2016. The majority were in response to inspection findings.
In 2016, CNSC staff conducted 1,452 inspections to verify compliance with CNSC regulatory requirements, including 228 security inspections to verify compliance against the requirements of REGDOC-2.12.3, Security of Nuclear Substances: Sealed Sources.
As part of CNSC staff efforts to consolidate compliance activities, the CNSC transferred compliance verification activities related to the import and export of high-risk sealed sources in the sectors covered in this report from the Directorate of Security and Safeguards to the Directorate of Nuclear Substance Regulation. In 2016, inspectors from the Directorate of Nuclear Substance Regulation took on this responsibility and inspected three licensees for their compliance to licence conditions related to the export of high-risk sealed sources. Two of the three were non-compliant with at least one of the requirements.
The CNSC continues to enhance the tools available to its inspectors. Recently this has included further integration of the Mobile Inspection Kit tablets into inspections, developing and updating available tools for performance-based inspections, and reviewing and updating inspector training and guidance documents to reflect current practices.
3.9 Stakeholder engagement
Clarity of requirements is one of the CNSC’s corporate priorities. Stakeholder engagement and outreach are two tools the CNSC uses to meet this priority. Outreach and engagement lead to an increased awareness and better understanding of the regulatory process and requirements. These, in turn, lead to increased workplace safety. CNSC staff takes all opportunities to perform outreach, including while on inspection.
Outreach sessions held throughout Canada in 2016 provided licensees and other persons the opportunity to interact with the regulator outside the scope of an inspection or licensing activities. Some of the key sessions are described below. In addition to outreach sessions, CNSC staff delivered presentations at various conferences in Canada and around the world to share information on developing regulatory topics.
3.9.1 Outreach sessions
Since 2009, the CNSC has offered an outreach program for licensees that use nuclear substances and prescribed equipment. The presentations made by CNSC staff and discussions associated with outreach are meant to inform licensees and other persons regulated by the CNSC on recent and upcoming regulatory changes, and provide education regarding the CNSC’s expectations for licensing and compliance requirements.
In 2016, the CNSC outreach program addressed recent and upcoming regulatory developments and other areas of regulatory focus, including inventory control and possession of disused sources, and upcoming changes to the Radiation Protection Regulations, specifically with regard to measuring doses to the eye. These outreach sessions were given in 15 cities across the country and were attended by more than 400 people.
3.9.2 Newsletters
In 2009, the CNSC introduced the DNSR Newsletter as an outreach vehicle for disseminating regulatory and safety information to licensees that use nuclear substances and prescribed equipment in Canada. The newsletter articles address various regulatory compliance issues and support the regulator’s commitment to keep both licensees and the public informed. Regular editions of the newsletter provide valuable information to licensees in all sectors; special editions focus on either a specific subsector or an area of regulatory interest.
All newsletters are posted on the CNSC website and are sent to recipients on the CNSC subscription list.
Three DNSR Newsletter editions were prepared in 2016: two regular editions and one special edition, which focused on measures for the security of sealed sources in accordance with the requirements and guidance indicated within REGDOC-2.12.3. Topics included in the regular editions of the newsletter covered CNSC’s support to first responders during the forest fires in Fort McMurray, a revision to the classes of nuclear substances for contamination control, clarification on the requirement for the retention of records for servicing work performed on radiation devices, highlights on relevant regulatory framework developments and upcoming consultation periods, and a summary of events reported to the Commission.
In May 2016, the CNSC published a special article to clarify CNSC expectations of licensees during skin contamination events. The article set a limit beyond which skin contamination must be reported to the CNSC, provided a flow chart to assist licensees in responding to skin contamination events, and offered guidance on how to calculate doses to the skin.
3.9.3 Industrial radiography working group
In 2009, a CNSC/industrial radiography working group was established to foster improved communications between the CNSC and the industry. The working group meets twice a year to discuss best practices and safety performance, and provides a forum in which stakeholders can stay informed of new developments from both technical and regulatory perspectives. In 2016, both meetings of the industrial radiography working group took place in Mississauga, Ontario. Discussions centered on items of interest to the industrial radiography community and planning an outreach strategy for the next annual meeting.
The CNSC holds two separate annual meetings with the radiography industry. In 2016, the meetings were held in Nisku, Alberta, and Ottawa, Ontario. The meeting in Nisku was attended by approximately 54 participants; the meeting in Ottawa was attended by 17 participants. CNSC staff use these meetings to address recent and upcoming regulatory developments and discuss other areas of regulatory focus. The meetings act as venues for industry members to communicate with CNSC staff, ask questions and share information on best practices and lessons learned. During the 2016 meetings, CNSC staff delivered presentations on common licensing roadblocks and security of nuclear sources, and provided updates on emergency situations and CNSC expectations for event reporting, compliance expectations during field inspections of industrial radiography licensees and the EDO certification program. Members of industry gave presentations to their peers on the basics of incident investigation.
3.9.4 Canadian Radiation Protection Association working group
In 2014, a working group was established between the CNSC and the Canadian Radiation Protection Association (CRPA). In 2016, this working group continued its efforts to promote strong radiation safety cultures within licensed activities.
For almost three decades, CNSC staff have delivered regulatory-focused presentations and participated in regulatory workshops at the CRPA’s annual conferences. At the 2016 conference in Toronto, Ontario, CNSC staff delivered presentations on radiation doses to the eye and the activation of air and concrete in cyclotron facilities.
3.9.5 Canadian Organization of Medical Physicists
The Canadian Organization of Medical Physicists (COMP) represents medical physicists working in radiotherapy facilities in the medical sector. Many certified radiation safety officers at Class II nuclear facilities are members of COMP.
CNSC staff attended the 2016 COMP Annual Scientific Meeting in St. John’s, Newfoundland and Labrador. CNSC staff delivered a presentation focused on the application of REGDOC-2.12.3, Security of Nuclear Substances: Sealed Sources in the context of Class II nuclear facilities in the medical sector. Tangible technical and administrative security measures were discussed, as was the importance of human behavior in ensuring the safety of nuclear substances.
3.9.6 CNSC-Class II/CRPA/COMP working group
The CNSC-Class II/CRPA/COMP (C3) working group was established in late 2015, with the mission of providing a forum for communication and information sharing among stakeholders in the regulated Class II community. The group met three times in 2016. Topics discussed included: quality control test frequencies for mandatory Class II safety systems, implementation of REGDOC-2.2.2, Personnel Training, version 2, promoting licensee internal and external peer audits as a mechanism for ensuring regulatory compliance, potential pathways for incorporating peer audits into the compliance verification framework, and knowledge versus competency as it pertains to the role of RSO.
3.9.7 Portable gauge workshops
Established in 2014, the CNSC regulatory workshop for portable gauge licensees was created to promote compliance and safety culture within this industrial subsector. The workshops consist of presentations delivered by CNSC staff, a question-and-answer session and general discussions. The presentations focus on radiation protection, compliance programs, worker training, transport of nuclear gauges and reporting requirements. CNSC staff developed this workshop in response to negative compliance trends along with the escalation of enforcement actions against licensees in this subsector.
CNSC staff offered 16 portable gauge workshops in 11 locations across Canada in 2016. Approximately 171 people attended these workshops. 2016 marked the final year of this initiative. CNSC staff are currently evaluating the program to determine if it should continue in its current format, or if other approaches will be used to target this subsector.
3.9.8 Nuclear medicine outreach
At the request of RSOs in the Montréal, Quebec region, CNSC staff participated in a half-day workshop targeting the nuclear medicine sector. Participants and CNSC staff discussed topics of interest to the nuclear medicine community, including items relating to skin contamination, the revision of the licence application guide, classification of nuclides and shipping empty packages. The workshop was attended by 30 RSOs.
4 Regulatory developments
This section provides details of the regulatory developments that took place in 2016 relating to regulatory programs for licensees covered in this report.
4.1 New licence conditions for nuclear substance and radiation device licensees
In 2016, three licence conditions applicable to nuclear medicine licensees were revised. These licence conditions only apply to licences issued by designated officers.
- LC 2583 – Patient Room Reassignment provides specific tasks to compete and criteria to meet regarding dose rates before a hospital room that was used by a patient that received a nuclear medicine treatment can be reassigned to a non-nuclear medicine patient. The revision added criteria pertaining to patient washrooms.
- LC 2110 – Area Classification – Nuclear Medicine was revised to clarify the requirements around classifying rooms or spaces in a hospital for nuclear medicine to include areas where nuclear substances used for nuclear medicine procedures can be prepared and administered and procedures performed.
- LC 2600 and LC 2601 – Thyroid Screening and Thyroid Bioassay were revised to expand the list of iodine isotopes, the uses of which require the worker to undergo thyroid screening procedures. The change was made reflect the increased use of various radioiodine isotopes within medical and academic and research sectors.
4.2 Guidance on the handling of the deceased implanted with nuclear substances
Following a request for clarification by stakeholders, CNSC staff have developed guidance for the handling of the deceased that have been implanted or injected with nuclear substances as well as those that have inhaled or ingested nuclear substances. The guidance is intended for coroners and funeral home and crematorium workers. It can be used when providing services to families of deceased patients who have undergone diagnosis or therapy using nuclear medicine or manual brachytherapy. It will inform readers of the risks that are present (minimal in all cases) and recommend best practices to help keep doses to the workers, families and the public at ALARA levels. REGDOC-2.7.3, Radiation Protection Guidelines for Safe Handling of Decedents will be published for consultation in 2017. In preparation, CNSC staff started conducting information sessions on the topic in 2016 to increase awareness about the forthcoming guideline, including presenting at the conference of the British Columbia Funeral Association in May 2016.
4.3 Regulatory focus in 2017
The CNSC’s focus in 2017 continues to be on effective regulatory oversight and continuous improvement. Activities will include:
- increased use of Type I inspections for compliance verification of large licensees
- clarifying expectations for reportable events with the development of regulatory document REGDOC-3.1.2, Part II, Reporting Requirements for Nuclear Substances and Radiation Devices
- enhanced oversight of RSOs across all sectors
- increased focus on preparing licensees with Category 3, 4 and 5 sealed sources to be compliant with the expectations of REGDOC-2.12.3, Security of Nuclear Substances: Sealed Sources, which will come into effect May 31, 2018
- augmented focus on compliance verification of fixed gauge licensees with the licence condition for vessel or hopper entry
- implementing regulatory oversight programs for new devices and technologies, particularly in the healthcare sector
- increased activity in certification of prescribed equipment and radiation devices due to the expiry of many existing certificates
5. Safety performance – all sectors combined
This section provides an overview of the overall performance of the industry sectors covered in this report.
5.1 Overall safety assessment
CNSC staff conducted 1,452 inspections across all sectors in 2016 to verify compliance with CNSC regulatory requirements, including 228 security inspections to verify new security requirements and three inspections related to the export of high-risk sources. All sectors continued to demonstrate adequate performance within all safety and control areas (SCAs). The majority of inspected licensees in 2016 were found to be compliant in the four SCAs covered in this report:
- In management system, 97.5 percent of licensees ensured that adequate processes and programs were in place to achieve their safety objectives.
- In operating performance, 87.4 percent of licensees made adequate provisions for the health, safety and security of persons, and protection of the environment.
- In radiation protection, 84.6 percent of licensees continued to ensure that exposure of workers and the public to ionizing radiation remained as low as reasonably achievable.
- In security, 93.4 percent of licensees demonstrated that they have adequate provisions in place to prevent the loss, sabotage, illegal use, illegal possession or illegal removal of sealed sources and prescribed equipment in their care and control.
For those in possession of high-risk sealed sources, security inspections were conducted in 2016 to measure compliance with the requirements of REGDOC-2.12.3, Security of Nuclear Substances: Sealed Sources. Of those inspected, 79 percent (181 of 228 inspections) were found to be compliant with the regulatory requirements. Licensees have put in place measures to correct all non-compliances identified during these inspections. The majority of non-compliances for security requirements were related to trustworthiness and reliability verification of employees, and licensee response protocols.
Effective doses to workers continued to be below regulatory limits in 2016, consistent with previous reporting years. Doses for 62,013 workers were reported to the CNSC in the four sectors covered in this report. Of those workers, 22,606 were designated as nuclear energy workers (NEWs) while 39,407 were not designated as NEWs.
One NEW received an equivalent dose above the regulatory limit for extremities of 500 millisieverets (mSv) as a result of an event that was reported to the Commission in December 2016. Further details on this event are provided in section 5.7.2.
A member of the public received a whole-body dose in excess of the regulatory limit after taking a ride from a ride-sharing website in a vehicle where the driver was also carrying packages containing nuclear substances. Further details are provided in section 5.7.6.
For 2016, CNSC staff assessed all 139 events reported by the licensees covered in this report. Reported events have been ranked using the International Nuclear and Radiological Event Scale (INES). Of these, 136 were ranked as level 0 (no safety significance), two were ranked as level 1 (anomaly) and one was ranked as level 2 (incident). The level 2 event involved the NEW who received a dose that exceeded the applicable regulatory limit mentioned above.
For all of the events reported, licensees implemented appropriate response measures to mitigate the impacts of the events and to limit radiation exposure to workers and the public. CNSC staff reviewed the measures put in place by licensees and found them to be satisfactory.
In 2016, six licensees received a rating of unacceptable in at least one SCA. The CNSC took escalated enforcement action in each case, issuing an order, an administrative monetary penalty (AMP) or both.
5.1.1 Licensees with compliance ratings below expectations
The CNSC takes a licensee’s compliance history into account when determining the inspection schedule. When a licensee repeatedly performs below expectations, the CNSC may increase the regulatory oversight of the licensee. This could include additional reactive inspections to monitor ongoing compliance or increased inspection frequency.
In 2016, 472 inspections did not meet compliance expectations in at least one SCA. The CNSC reviewed past performances of these licensees and noted that 12 percent of these same licensees were rated below expectations or unacceptable in that same SCA on their last inspections. This is consistent with industry performance in general, however the CNSC will track trends in this area in future editions of this report to assess trends.
5.2 Management system
The management system SCA covers the framework that establishes the processes and programs required to ensure that an organization achieves its safety objectives, continuously monitors its performance against those objectives, and fosters a healthy safety culture.
All sectors demonstrated satisfactory performance within the management system SCA, with 97.5 percent of inspected licensees (1,259 of 1,290 inspections) found to be in compliance with regulatory requirements (see figure 4). A breakdown of the inspection ratings by sector for 2016 is shown in figure 5.
The majority of non-compliances in this SCA included licensees failing to notify the CNSC of changes to their radiation safety officers (RSOs) or applicant authorities within the allotted timelines, failing to keep required records at all work locations, including any temporary work locations, and conducting activities for which they were not licenced (generally, these tasks were administrative in nature such as using nuclear substances not listed on their licences or possessing radiation device models that did not appear on their licences).
.png/object)
Description
| Rating | 2015 | 2016 |
|---|---|---|
| Fully satisfactory or satisfactory | 1,233 | 1,261 |
| Below expectations | 49 | 27 |
| Unacceptable | 0 | 2 |
.png/object)
Description
| Sector | 2015 | 2016 |
|---|---|---|
| Medical | 227 | 207 |
| Industrial | 837 | 901 |
| Academic and research | 67 | 73 |
| Commercial | 102 | 80 |
| All sectors combined | 1,233 | 1,261 |
5.3 Operating performance
Operating performance refers to the licensee’s ability to perform licensed activities in accordance with pertinent operational and safety requirements defined in the Nuclear Safety and Control Act (NSCA), its associated regulations and licence conditions. Licensees are expected to demonstrate that they comply with operational and safety requirements by providing workers with appropriate procedures for the safe use of nuclear substances and prescribed equipment, by ensuring that workers follow procedures, and by maintaining records that demonstrate compliance.
All sectors continued to demonstrate adequate performance within the operating performance SCA in 2016, with 87.4 percent of inspected licensees (1,147 of 1,313 inspections) found to be in compliance with regulatory requirements. This is down slightly from 2015 when 90.6 percent of licensees were in compliance.
Five inspections resulted in a rating of unacceptable for the operating performance SCA in 2016. In all cases, inspectors issued orders to the licensees to stop the unsafe work practices and ensure corrective actions were taken immediately. Compliance with regulatory requirements is unacceptable when compliance within the overall SCA is significantly below expectations, or there is evidence of systemic failure in safely conducting activities. Without immediate corrective action, there is a high probability that the deficiencies will lead to an unreasonable risk.
Inspection ratings for all sectors combined are shown in figure 6 and a sector-to-sector comparison appears in figure 7. The academic and research sector showed significant improvement in this SCA in 2016 compared to 2015. It has rebounded from the downward trend it had been following since 2013. All other sectors experienced a slight decrease in the SCA rating year-over-year, but remained within the range observed over the past five years.
The most common non-compliances in this SCA included failure to comply with regulatory requirements related to workers’ responsibilities to following licensee procedures and use equipment provided by the licensee, following procedures in the documents appended to the licence, and keeping training records of employees.
.png/object)
Description
| Rating | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Fully satisfactory | 1,308 | 1,400 | 1,260 | 1,181 | 1,147 |
| Below expectations | 179 | 166 | 164 | 120 | 161 |
| Unacceptable | 5 | 4 | 1 | 2 | 5 |
.png/object)
Description
| Sector | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Medical | 186 | 271 | 191 | 229 | 201 |
| Industrial | 781 | 839 | 839 | 785 | 792 |
| Academic and research | 223 | 178 | 118 | 60 | 74 |
| Commercial | 118 | 112 | 112 | 107 | 80 |
| All sectors combined | 1,308 | 1,400 | 1,260 | 1.181 | 1,147 |
5.4 Radiation protection
Radiation protection programs are required for every licensee to ensure that contamination levels and radiation doses received by workers are monitored, controlled and maintained below regulatory dose limits, and kept at levels that are as low as reasonably achievable (ALARA), social and economic factors being taken into account. Licensees are expected to monitor worker doses, post radiation warning signs, plan appropriately for radiological emergencies, manage oversight of operational activities, institute effective workplace practices that emphasize the use of time, distance and shielding to minimize exposure to radiation, and use appropriate protective equipment
.png/object)
While all sectors demonstrated adequate performance within this SCA, with 84.6 percent of inspected licensees (1,108 of 1,311 inspections) found to be compliant with regulatory requirements, this figure represents a lower level of compliance than has been observed since 2013. (See figure 9).
In 2016, four inspections resulted in unacceptable ratings for radiation protection. Three of these inspections also had unacceptable ratings for the operating performance SCA. In all four cases, the CNSC inspectors issued an order to immediately stop the unsafe work practices and to implement corrective measures.
Inspection ratings for all sectors combined are shown in a sector-to-sector comparison presented in figure 10. After gains made in performance in this SCA up to 2014, the medical sector performance in this SCA has decreased over the past two years.
The majority of non-compliances included failing to use calibrated radiation survey meters, failing to implement radiation programs that keep doses to workers and the public ALARA, and failure to post radiation warning signs as required.
.png/object)
Description
| Rating | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Fully satisfactory or satisfactory | 1,265 | 1,351 | 1,269 | 1,151 | 1,108 |
| Below expectations | 237 | 200 | 152 | 146 | 199 |
| Unacceptable | 10 | 4 | 4 | 0 | 4 |
.png/object)
Description
| Sector | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Medical | 152 | 234 | 192 | 212 | 185 |
| Industrial | 771 | 833 | 842 | 768 | 773 |
| Academic and research | 213 | 174 | 117 | 65 | 71 |
| Commercial | 129 | 110 | 118 | 106 | 79 |
| All sectors combined | 1,265 | 1,351 | 1,269 | 1,151 | 1,108 |
5.5 Security
Licensees are required to have in place physical security measures, practices and programs to prevent the loss, illegal use, illegal possession or illegal removal of nuclear substances during their entire lifecycle, including while they are in storage or during transport, as per the NSCA. The extent of the security measures required depends upon the types of nuclear substances used and activities performed by each licensee.
Overall, all sectors showed satisfactory ratings for the security SCA in 2016: 93.6 percent of inspected licensees (1,152 of 1,234 inspections) were compliant with regulatory requirements. One inspection resulted in a rating of unacceptable. The inspector issued an order to ensure corrective actions were taken immediately.
Licensees with high-risk sources are subject to the requirements described in REGDOC-2.12.3, Security of Nuclear Substances: Sealed Sources. In 2016, CNSC inspectors conducted security inspections to verify compliance against those requirements. Of those inspected, 79 percent (181 of 228 inspections) were found to be compliant with regulatory requirements applicable to Category 1 and 2 sealed sources. Generally, the licensees have in place basic physical security measures. Non-compliances are usually for administrative requirements introduced with REGDOC-2.12.3, including implementation of a trustworthiness and reliability verification program, alarm testing, and event-response procedures.
Licensees addressed and corrected all non-compliances identified during inspections to the satisfaction of the CNSC. Figure 11 summarizes the performance of all sectors combined for this SCA for 2014 to 2016, while figure 12 provides a sector-to-sector comparison for those three years.
.png/object)
Description
| Rating | 2014 | 2015 | 2016 |
|---|---|---|---|
| Fully satisfactory or satisfactory | 1,265 | 1,149 | 1,155 |
| Below expectations | 67 | 59 | 78 |
| Unacceptable | 0 | 2 | 1 |
.png/object)
Description
| Sector | 2014 | 2015 | 2016 |
|---|---|---|---|
| Medical | 181 | 219 | 190 |
| Industrial | 875 | 780 | 830 |
| Academic and research | 120 | 64 | 70 |
| Commercial | 89 | 86 | 65 |
| All sectors combined | 1,265 | 1,149 | 1,155 |
Compliance with the mandatory tracking of high-risk sealed sources was satisfactory in 2016. Of the 150 inspected licensees, 141 (94 percent) were found to be compliant with this requirement. CNSC staff ensured that all instances of non-compliances were adequately addressed by the licensees. The majority of non-compliances for high-risk sealed source tracking requirements were administrative items such as failing to provide notification of shipments or receipts within specified time frames, failing to register location with the CNSC, or providing incorrect location identification for devices or sources. Further information on this topic is available in the National Sealed Source Registry and Sealed Source Tracking System Annual Report.
5.6 Enforcement actions
In 2016, the CNSC took 22 escalated enforcement actions against licensees in the medical, industrial, academic and research, and commercial sectors (figure 13). CNSC staff issued orders directing licensees to take immediate corrective measures in 14 instances. In each case, the licensee immediately complied with the order. Once the CNSC was satisfied that the licensee had addressed the order’s terms and conditions, the order was closed. All orders issued in 2016 are closed. CNSC designated officers issued administrative monetary penalties (AMPs) in eight instances in 2016, all of which have been paid. A breakdown of the 22 enforcement actions is provided for each of the sectors while a summary of orders and AMPs issued by the CNSC in 2016 is provided in appendix C. Further information on regulatory actions, including escalated enforcement actions, taken by the CNSC is also available on the CNSC website.
.png/object)
Description
| Sector | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Medical | – | – | – | 2 | 1 |
| Industrial | 16 | 24 | 16 | 14 | 18 |
| Academic and research | – | – | 2 | 1 | – |
| Commercial | 1 | – | 1 | 4 | 3 |
| All sectors combined | 17 | 24 | 19 | 21 | 22 |
5.7 Reported events
Licensees are required to have programs in place for the management of unplanned events and accidents. The events that warrant mandatory reporting and the content of the reports are set out in the NSCA, its regulations and the conditions of their licence. CNSC staff review, assess and track all events reported by licensees.
Since 2014, reported events have been ranked using the International Nuclear and Radiological Event Scale (INES), a tool for communicating the safety significance of nuclear and radiological events to the public. This tool allows the establishment of a proper perspective of an event’s safety significance. The scale has been used to classify events at nuclear power plants since 1990 and has been extended over the years to apply to all nuclear industry installations. By 2006, it had been adapted to all events associated with the transport, storage and use of radioactive sources and nuclear substances. Note that the scale is not a tool to compare safety performances among facilities or organizations, but to effectively communicate the safety significance of events.
In 2016, there were 139 events related to nuclear substances reported to the CNSC by licensees in the sectors covered in this report. Of these events, 136 were ranked as INES level 0 (no safety significance) and two were ranked as level 1 (anomaly) based on the quantity of nuclear substances involved and the type of event reported (stolen portable gauges). The remaining event – ranked as level 2 (incident) – occurred when a spill resulted in a NEW received an extremity dose of 1,100 mSv, which is above the regulatory limit of 500 mSv. This event was reported to the Commission on December 14, 2016.
A breakdown of reported events by type is shown in figure 14 and a complete list of all reported events in 2016 is provided in
appendix D.
For all of the events reported, the licensees implemented adequate response measures to mitigate the impacts of the events and to limit radiation exposure to workers or any radiological impact on the environment. CNSC staff reviewed these measures, along with licensee corrective actions to prevent recurrence of the events, and found them to be satisfactory.
As part of their final, detailed reports on events, licensees are required to identify probable causes of events and propose corrective actions to prevent recurrences. In many cases, probable causes were related to workers not following procedures. As a result, the majority of measures taken by licensees to prevent recurrence related to retraining staff on procedures and emphasizing the importance of procedural adherence.
In addition to the events related to the use of nuclear substances, there was one occurrence of an injured worker. The licensee reported that a portable gauge operator was injured when he was pinned under his truck at a work site. He was taken to hospital.
.png/object)
Description
| Events | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Malfunctioning or damaged devices | 52 | 51 | 52 | 53 | 44 |
| Spills, contamination or release | 31 | 39 | 39 | 17 | 20 |
| Missing or found nuclear substances | 18 | 17 | 17 | 20 | 13 |
| Packaging and transport | 27 | 26 | 30 | 47 | 44 |
| Breach of security | 11 | 17 | 9 | 5 | 5 |
| Unplanned exposure | – | – | – | 13 | 12 |
| All reported events | 139 | 150 | 147 | 155 | 139 |
Note: Note: Unplanned exposure represents events that have led to non-routine exposures to workers or members of the public. Events of this type were covered under breach of security prior to 2015.
5.7.1 Malfunctioning or damaged devices
There were 44 events related to damaged or malfunctioning devices. Of these, 25 were reported as damaged devices:
- Twelve involved damage to portable gauges. In one case, the gauge was damaged when it fell off the back of a truck. In the remaining cases, damage occurred when the portable gauges were hit or run over by vehicles and equipment at construction sites.
- Two involved damage to fixed gauges. In one case, gauges were damaged by fire.
- Seven involved damage to exposure devices, often following a drop or an impact.
- One involved the crank of an exposure device that was damaged after coming into contact with a hot surface.
- One involved a radiation device damaged when hit by a crane.
- Two involved damaged devices found in storage. One device was a portable gauge, the other an exposure device.
None of the above resulted in damage to the source, or resulted in source leakage.
One licensee reported damaging a sealed source when a radiotherapy seed was cut in half during preparation of a tissue sample. There was no release to the environment or exposure to members of the public as a result of this event.
The remaining 19 events in this category corresponded to malfunctioning devices:
- Ten involved malfunctioning fixed gauges where the shutters failed to close properly or the indicators for the shutters were not functioning properly.
- Six involved malfunctioning portable gauges where the shutters were stuck in the open position.
- Two involved malfunctioning exposure devices where the sealed sources failed to retract into the shielded position.
- One involved a malfunctioning gamma checker gauge where there were problems with the source control crank. This device is used to expose survey meters to known and reproducible radiation fields.
All malfunctioning devices were taken out of service as required by the Nuclear Substances and Radiation Devices Regulations and either repaired or sent for disposal. Any radiation exposure to a person as a result of these events was well below the annual public regulatory dose limit of 1 mSv. All events in this category are closed.
5.7.2 Spill or contamination
All licensees are required to document, record and investigate every skin contamination event to ensure work practices are optimized and to minimize the probability of repeat occurrences. None of the spill, contamination or release events reported in 2016 posed a risk to the environment or resulted in members of the public receiving a dose.
There were 20 events related to spills or minor contamination of personnel reported:
- Eight incidents involved spills of technetium-99m. Three of these resulted in skin contamination of workers. In one case, the worker was able to wash off the contamination. In the remaining two cases, the workers were unable to completely remove the contamination by washing their hands, resulting in a small extremity dose well below the regulatory limits.
- One incident involved a spill of indium-111 without contamination to a person.
- There were four spills involving fluorine-18. Three resulted in contamination of the skin of the workers involved. In all three cases, the contamination was quickly removed and did not result in the workers receiving doses above regulatory limits.
- One incident involved a spill of strontium-82 without contamination to a person.
- Four spills involved iodine-131 without contamination to a person.
- Two spills involved yttrium-90. One of the incidents resulted in contamination of a worker and a dose to the skin of his hands above the regulatory limit of 500 mSv. This event was presented to the Commission on December 14, 2016.
5.7.3 Missing, stolen or found nuclear substances
Licensees are required to have in place physical security measures, practices and programs to prevent the loss, illegal use, illegal possession or illegal removal of nuclear substances during their entire lifecycle, including while they are in storage or during transport.
In 2016, there were 10 reported events involving lost or stolen nuclear substances.
- Nine events involved Category 3, 4 and 5 sources; considered to be of moderate, low and very low risk respectively. The sealed sources or radiation devices were recovered in four of these cases.
- One event involved a Category 2 source (high risk). A licensee lost an exposure device while transporting it to a job site. The licensee recovered the device the following day. Details of this event were reported to the Commission on August 18, 2016.
In 2016, one portable gauge stolen in 2015 was recovered.
In 2016, there were two events in which nuclear substances were found in the public domain. One was a low-risk device (i.e., a dew point analyzer) containing radium-226, which is a Category 5 sealed source. The second item was an unidentified piece of metal with radiation markings and radiation levels above background. Both devices were found at scrap metal facilities. Inspectors from the CNSC took possession of the devices and will make arrangements for their disposal.
Events involving lost stolen or recovered radiation devices and sealed sources are reported in the Lost or Stolen Sealed Sources and Radiation Devices Report, which is updated regularly. Table 4 provides a summary of the events involving missing and found nuclear substances that occurred in 2016.
| # | Date | Event summary | Sealed source category | INES rating level | Status |
|---|---|---|---|---|---|
| 2677 | January 8 | A licensee reported sealed sources as lost when he was unable to account for the exact location of the sources. | 5 | 0 | Not recovered (very low risk) |
| 2760 | May 23 | A vehicle containing a portable gauge was reported stolen. The vehicle and the portable gauge were recovered. | 4 | 0 | Recovered |
| 2769 | June 15 | A therapeutic dose of iodine-131 was reported as missing following delivery. | n/a | 0 | Not recovered (decayed) |
| 2775 | June 28 | A dew pointer was found in a load of scrap metal. | 5 | 0 | Found source |
| 2793 | August 3 | A licensee reported the loss of an exposure device while transporting it to a job site. The exposure device was recovered the following day. | 2 | 0 | Recovered |
| 2823 | September 9 | A portable gauge was stolen from a construction site. The police were notified and the portable gauge was recovered. | 4 | 0 | Recovered |
| 2866 | October 4 | A car transporting a portable gauge was reported as missing. It was determined that the car had been towed to a city impound lot. The device and car were recovered. | 4 | 0 | Recovered |
| 2867 | October 4 | A portable gauge was stolen from a car parked at a private residence. The device has not been recovered. | 4 | 0 | Not recovered (low risk) |
| 2869 | October 4 | A piece of metal with a radiation warning sign was discovered at a scrap metal facility. | 5 | 0 | Found source |
| 2883 | October 21 | A nickel-63 sealed source was discovered missing from a gas chromatograph. The source was recovered. | 5 | 0 | Recovered |
| 2627 | November 10 | A portable gauge stolen on November 7, 2015 was recovered. | 4 | 0 | Recovered |
| 2921 | December 2 | A portable gauge was stolen from a truck parked outside a private residence. The device has not been recovered. | 4 | 1 | Not recovered (low risk) |
| 2925 | December 21 | A Category 5 source (i.e., a cancer seed) was reported as lost. | 5 | 0 | Not recovered (very low risk) |
5.7.4 Breach of security
The extent of the security measures required by a licensee depends upon the types of nuclear substances used and activities conducted. In 2016, there were five events reported to the CNSC relating to breaches of security.
- One involved the discovery of an unlocked room with the intrusion alarm unarmed during a security sweep of a secure nuclear medicine area. The licensee’s investigation determined that no nuclear substances were missing. The licensee determined that a new member of the housekeeping staff was given access to the room by the radiotherapy manager who failed to inform the staff member of the requirement to arm the alarm system or lock the door once cleaning of the room was complete. The licensee addressed the training and communication issues that led to this event. These measures were reviewed by CNSC staff and found to be satisfactory.
- One involved the theft of keys for a room that contained prescribed equipment. The licensee added extra security surveillance to the area until the locks were rekeyed. There was no unauthorized access to the prescribed equipment or prescribed information.
- One involved an unauthorized person working in a laboratory area dedicated for work with nuclear substances at a university. The licensee’s investigation determined that the individual was not working with nuclear substances. The licensee revised its procedure for visitors in laboratories. The measures were reviewed by CNSC staff and found to be satisfactory.
- One involved a break-in at licensee’s location used for the storage of portable nuclear gauges. This was the second time that this location was targeted by thieves. The locker containing the portable gauges had not been tampered with and no devices or prescribed information were stolen. As a result, the licensee put in place heightened security measures at this location. A third break-in occurred in early 2017. In response, CNSC staff met with the local police to discuss the situation, and conducted reactive security inspections at this location as well as at other locations operated by this licensee to verify that security measures in place were adequate.
- One involved an access card to a restricted area that was found in a public washroom at a licensee’s facility. Security staff immediately deactivated the card, and then verified and confirmed that the card had not been used. The licensee modified its procedures to reduce the likelihood of a similar event in the future.
There was no access to, or theft of, nuclear substances or radiation devices as a result of any of these events. All of the events are closed.
5.7.5 Packaging and transport
Approximately one million packages containing nuclear substances are safely transported each year in Canada. In 2016, there were 44 events reported to the CNSC relating to packaging and transport.
- Sixteen were accidents involving vehicles transporting nuclear substances or radiation devices that did not resulted in any damage to the packages on board.
- Eleven involved packages that were misrouted or delayed during transport. In all cases, the packages were delivered to the proper location or returned to the originator.
- Ten incidents involved packages that were damaged during transport. In all cases, the reports concluded that the nuclear substances or radiation devices were fully contained within the packages and that there was no release as a result of the incidents.
- Two involved packages with external contamination following transportation. In both cases, the licensees isolated the package to prevent further contamination of workspaces and left the packages to decay. The contamination levels were low and posed no risk to the workers who received the packages.
- One involved a package that was not labelled in accordance with the Packaging and Transport of Nuclear Substances Regulations, 2015. The package contained nuclear substances, but was not labelled accordingly.
- One involved improper packaging of a source. The source was not inserted into the shielded position, which led to a dose rate on the surface of the package that was higher than expected, but still within regulatory limits.
- One involved a package left behind in a public area. The package was retrieved by licensee staff. No members of the public were affected by the event.
- One involved a package that was not classified in accordance with the Packaging and Transport of Nuclear Substances Regulations, 2015. The package was classified as an excepted package for transport instead of being classified as a Type A package. The packaging used for transportation met all the requirements applicable to Type A packages.
- One involved a vial of fluorine-18 that was broken upon opening of the package. The interior of the package was contaminated, however there was no contamination on the exterior of the package.
None of the events resulted in releases to the environment or doses to members of the public above regulatory limits.
5.7.6 Unplanned exposures to persons
When nuclear substances are used, there may be situations that lead to unplanned exposures to persons. Often these events involve people entering restricted work areas, such as required in the industrial radiography subsector.
In 2016, there were 13 events reported to the CNSC that led to unplanned exposures to persons. Twelve of these were reported by licensees and one was reported through the CNSC public complaints process. The event reported through the CNSC public complaints process is the only event that led to a dose above a regulatory dose limit. All events are closed.
- Six events involved breaches of safety barriers where workers not involved in the operation of radiation devices or Class II prescribed equipment entered restricted areas that were established prior to the use of exposure devices or linear accelerators. In all cases the workers received doses below the public regulatory dose limit of 1 mSv.
- Three events involved contract workers who entered vessels when the shutter of the fixed gauges installed inside the vessels were left in the open position. In two events, the workers were employees of the licensee but were non-NEWs. The third event involved a contract worker who was also a non-NEW. In all cases, the individuals received doses below the public dose limit of 1 mSv. In all three cases, the licensee used procedures that were not compliant with a condition of the licence pertaining to vessel entries. Administrative monetary penalties were issued to two of the three licensees as a result of the potential overexposures.
- Two events involved NEWs who were pricked with needles during nuclear medicine procedures. One NEW was exposed to iodine-131 and the other to fluorine-18.
- One involved a NEW who was contaminated with technetium-99m during the preparation of nuclear medicine doses. The estimated dose received was below regulatory limits.
The final occurrence of an unplanned exposure was brought to the attention of the CNSC staff through the CNSC’s public complaints program. It was not reported by a licensee.
- A member of the public received a dose in excess of the annual effective dose limit of 1 mSv/year. The member of the public accepted, along with four other members of the public, a ride advertised on a travel-share website from a driver offering to drive passengers at the same time he was delivering packages containing nuclear substances. This practice is not authorized under the Packaging and Transport of Nuclear Substances Regulations, 2015. Based on initial information, the CNSC made a conservative estimate that the passenger most affected as result of a 10-hour trip would have received an effective dose of approximately 1.62 mSv. CNSC staff informed the affected person of the effective dose received and the consequences of the radiation dose they received. The carrier terminated the driver and modified its internal procedures to include internal unannounced audits at drop-off locations. The CNSC issued an administrative monetary penalty to the driver of the vehicle as a result of this event in 2017. This event was reported to the Commission on December 14, 2016.
5.7.7 International events in 2016
The CNSC monitors events reported to the International Atomic Energy Agency (IAEA) through the IAEA’s International Reporting System for Operating Experience, a database of events that occur around the world. Regulators report events that occur in their countries on a voluntary basis for the purpose of sharing information. Reported events are assigned a significance according to the INES scale. In 2016, eight level 2 events involving nuclear substances and radiation devices were reported to the IAEA, which are summarized in the spring 2017 edition of the DNSR Newsletter.
These events include:
- An exposure device operator received a dose to his finger above the regulatory limit while repairing a guide tube and the source did not return to the shielded position.
- Workers were conducting industrial radiography work in an area with a known high radiation field. The workers ignored their personal dosimeter alarms and continued working, with a potential for very high doses to the workers.
- An industrial radiography worker working too close to the source received an effective whole body dose of 55 mSv.
- A 1.3 terabecquerel (TBq) selenium-75 source was damaged during the repair of industrial radiography equipment. Contamination was spread outside the controlled area and members of the public received doses slightly above the regulatory limit.
- Twenty members of the public received doses above regulatory limits when a 16-year old boy found a sealed source used for industrial radiography and took the source home.
- A sealed source used for industrial radiography was transported in what was marked as an empty package and transported with general luggage on a passenger flight.
- A nuclear pharmacist received a dose to his hand of 511 mSv in a year, which was above the regulatory limit. An investigation determined the individual was not following procedures for working with radiation sources at a distance.
- A cyclotron was operated while the bunker door was open. Workers were present at the bunker location for about four minutes before they became aware of the situation.
5.7.8 Response to forest fires in Fort McMurray
In the spring of 2016, a forest fire forced the evacuation of Fort McMurray, Alberta. At the time of the fire, there were more than 40 CNSC-licensed locations in and around Fort McMurray. CNSC staff contacted affected licensees to verify the status of storage locations, and provided a list of the locations where nuclear substances and radiation devices were stored with the Alberta Disaster Response Team. At the request of the Alberta Emergency Management Agency’s Provincial Operations Centre, two CNSC staff were dispatched to assess the potential risk to the public and the environment as a result of the forest fire. Based on the storage locations and validation by field verifications, CNSC staff concluded that all of the locations were unaffected by the forest fires. A detailed presentation on the CNSC response to the fire in Fort McMurray was provided at the Commission meeting held on June 22, 2016.
5.8 Effective doses to workers
A total of 62,013 workers in the four nuclear sectors covered in this report were monitored for occupational doses in 2016, of which 22,606 were designated as NEWs.
One NEW from the medical sector exceeded the annual regulatory extremity dose limit of 500 mSv for the hands. More details on this event can be found in section 5.7.2
Figure 15 shows the dose distribution for all workers in 2016. All workers that received doses above 1 mSv in 2016 were NEWs.
.png/object)
Description
| Sector | ≤ 0.5 | > 0.5 and ≤ 1 mSv | > 1 and ≤ 5 mSv | > 5 and ≤ 20 mSv | > 20 and ≤ 50 mSv | > 50 mSv |
|---|---|---|---|---|---|---|
| Medical | 7,977 | 581 | 1,181 | 63 | – | – |
| Industrial | 38,003 | 2,880 | 1,851 | 337 | 1 | – |
| Academic and research | 7,137 | 57 | 41 | 5 | – | – |
| Commercial | 1,522 | 145 | 212 | 19 | 1 | – |
The differences in doses to workers among sectors reflect the nature of the various activities within those sectors figure 16 shows the doses received by the 22,606 NEWs monitored in 2016 while figure 17 shows the doses of NEWs from 2012 to 2016.
.png/object)
Description
| Sector | ≤ 0.5 | > 0.5 and ≤ 1 mSv | > 1 and ≤ 5 mSv | > 5 and ≤ 20 mSv | > 20 and ≤ 50 mSv | > 50 mSv |
|---|---|---|---|---|---|---|
| Medical | 4,728 | 532 | 1,180 | 63 | – | – |
| Industrial | 7,944 | 1,922 | 1,851 | 337 | 1 | – |
| Academic and research | 2,575 | 49 | 41 | 5 | – | – |
| Commercial | 1,014 | 132 | 212 | 19 | 1 | – |
.png/object)
Description
| Year | ≤ 0.5 | > 0.5 and ≤ 1 mSv | > 1 and ≤ 5 mSv | > 5 and ≤ 20 mSv | > 20 and ≤ 50 mSv | > 50 mSv |
|---|---|---|---|---|---|---|
| 2012 | 7,882 | 820 | 1,446 | 157 | – | – |
| 2013 | 16,918 | 1,745 | 3,223 | 507 | 12 | – |
| 2014 | 18,044 | 1,719 | 3,420 | 499 | 6 | – |
| 2015 | 17,272 | 1,518 | 3,028 | 501 | 3 | – |
| 2016 | 16,261 | 2,635 | 3,284 | 424 | 2 | – |
Note: For 2012, the doses provided were based on only a representative sample of annual compliance reports within each sector.
6 Medical sector
Licensees in the medical sector use nuclear substances and operate accelerators and other Class II prescribed equipment for diagnostic and therapeutic purposes in hospitals and medical clinics. In 2016, the medical sector accounted for 470 CNSC licences and 9,802 total workers, of which 6,503 were designated as nuclear energy workers (NEWs).
The results of CNSC staff evaluation of the regulatory performance of all medical sector licensees inspected in 2016 are included in the overall results. The following three subsectors are highlighted in further detail:
- nuclear medicine
- radiation therapy
- veterinary nuclear medicine
.png/object)
6.1 Sector overview
Medical applications using radiopharmaceuticals target specific tissues and organs, and allow for the delivery of nuclear substances to specific areas of the body for diagnostic testing or treatment.
Diagnostic nuclear medicine studies assist in the diagnosis of medical conditions based on the physiological functions of organs, tissues or bones. Radiopharmaceuticals containing nuclear substances such as technetium-99m and fluorine-18 are administered to patients for imaging purposes. Examples of common diagnostic nuclear medicine procedures include myocardial perfusion scans (to visualize heart function and blood flow), bone scans (to evaluate bone metabolism, infection or tumours) and renal scans (to evaluate kidney function).
Radioisotopes are also used in many therapeutic nuclear medicine procedures. For example, iodine-131 is used to treat diseases of the thyroid gland, while other isotopes such as yttrium-90 may be used in conjunction with antibodies for site-specific treatment of certain cancers.
Medical linear accelerators and brachytherapy equipment are also used for therapeutic procedures. These devices are used to treat cancer by delivering carefully controlled doses of radiation to cancerous tissue.
Veterinary nuclear medicine uses techniques similar to those employed in human nuclear medicine. Veterinary clinics across the country offer a wide range of diagnostic and therapeutic nuclear medicine procedures and, in some cases, radiation therapy treatment using medical accelerators.
.png/object)
6.2 Summary of safety assessment
Based on their evaluation and verification of licensee performance, CNSC staff concluded that the safety performance of the medical sector was satisfactory in 2016.
Doses received by NEWs in this sector remained low, with the majority of workers receiving effective doses below 1 millisievert (mSv). One NEW received a dose to the hands above the annual limit.
Of the inspected licensees in 2016, the majority were found to be compliant in the four safety and control areas (SCAs) covered in this report:
- 95.8 percent were compliant in management system
- 88.2 percent were compliant in operating performance
- 80.2 percent were compliant in radiation protection
- 85.6 percent were compliant in security
In cases where non-compliances were noted, licensees took appropriate corrective actions, satisfactory to CNSC staff, to address the non-compliances.
The CNSC issued one administrative monetary penalty (AMP) in the medical sector in 2016 for non-compliances related to inventory control of sealed sources.
6.3 Safety performance measures
6.3.1 Doses to workers
NEWs in the nuclear medicine subsector continued to receive higher doses than workers in other medical subsectors as a result of directly administering nuclear substances to patients and constantly working in environments where patients are in close proximity to health professionals. The vast majority of these NEWs received doses below 5 mSv, as shown in figure 20. The doses to NEWs in the nuclear medicine subsector over the period of 2012 to 2016 are shown in figure 21.
In 2016, one NEW in the medical sector received a dose to the skin of the hands above the regulatory limit of 500 mSv. Details of this event are presented in section 5.7.2.
.png/object)
Description
| Sector | ≤ 0.5 | > 0.5 and ≤ 1 mSv | > 1 and ≤ 5 mSv | > 5 and ≤ 20 mSv | > 20 and ≤ 50 mSv | > 50 mSv |
|---|---|---|---|---|---|---|
| Nuclear medicine | 1,675 | 506 | 1,143 | 56 | – | – |
| Radiation therapy | 2,545 | 7 | 7 | 1 | – | – |
| Veterinary nuclear medicine | 122 | 7 | 1 | – | – | – |
| Medical sector | 4,728 | 532 | 1,180 | 63 | – | – |
Note: The total number of NEWs shown in the medical sector row is the aggregate for the entire sector, including subsectors not highlighted in this report.
.png/object)
Description
| Year | ≤ 0.5 | > 0.5 and ≤ 1 mSv | > 1 and ≤ 5 mSv | > 5 and ≤ 20 mSv | > 20 and ≤ 50 mSv | > 50 mSv |
|---|---|---|---|---|---|---|
| 2012 | 816 | 292 | 790 | 65 | N/A | N/A |
| 2013 | 1,531 | 458 | 1,236 | 56 | N/A | N/A |
| 2014 | 1,747 | 510 | 1,529 | 56 | N/A | N/A |
| 2015 | 1,679 | 477 | 1,205 | 40 | N/A | N/A |
| 2016 | 1,675 | 506 | 1,143 | 56 | – | – |
Note: For 2012, the doses provided were based on only a representative sample of annual compliance reports within each sector.
6.3.2 Management system
The overall compliance rating for management systems in the medical sector was 95.8 percent (207 of 216 inspections) in 2016. As shown in figure 22, the majority of licensees inspected by the CNSC were found to be compliant. A sector-to-subsector comparison of inspection ratings is provided in figure 23. The radiation therapy licensees showed low compliance ratings in this SCA. This phenomenon is amplified by the small number of licensees. The main non-compliances exhibited by radiation therapy licensees in this SCA were administrative in nature, and related to maintaining records and reviewing policies and procedures within the required timelines. The primary non-compliances for other medical licensees for this SCA were: using nuclear substances not included on their licences, failing to notify the CNSC of changes to the hospital names, and failing to notify the CNSC of changes to radiation safety officer and applicant authorities.
.png/object)
Description
| Inspection ratings | 2015 | 2016 |
|---|---|---|
| Fully satisfactory or satisfactory | 227 | 207 |
| Below expectations | 15 | 9 |
| Unacceptable | 0 | 0 |
.png/object)
Description
| Sector | 2015 | 2016 |
|---|---|---|
| Nuclear medicine | 189 | 167 |
| Radiation therapy | 13 | 7 |
| Veterinary nuclear medicine | 6 | 9 |
| Medical sector | 227 | 207 |
Note: The number of inspections shown in the medical sector row is the aggregate for the entire sector, including subsectors not highlighted in this report.
6.3.3 Operating performance
The overall compliance rating for operating performance in the medical sector was 88.2 percent (201 of 228 inspections) in 2016. As shown in figure 24, the majority of licensees inspected by the CNSC were found to be compliant. A sector-to-subsector comparison of inspection ratings is provided in figure 25.
The most common non-compliances in the operating performance SCA were workers not following procedures, or not using the equipment provided to them by the licensees, and licensees not following the procedures written in their radiation safety manuals.
.png/object)
Description
| Inspection ratings | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Fully satisfactory or satisfactory | 186 | 271 | 191 | 229 | 201 |
| Below expectations | 19 | 26 | 18 | 17 | 27 |
| Unacceptable | 0 | 0 | 0 | 0 | 0 |
.png/object)
Description
| Inspection ratings | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Nuclear medicine | 151 | 216 | 146 | 191 | 159 |
| Radiation therapy | 5 | 25 | 18 | 13 | 22 |
| Veterinary nuclear medicine | 6 | 6 | 8 | 6 | 9 |
| Medical sector | 186 | 271 | 191 | 229 | 201 |
Note: The number of inspections shown in the medical sector row is the aggregate for the entire sector, including subsectors not highlighted in this report.
6.3.4 Radiation protection
The overall compliance rating for radiation protection in the medical sector was 80.1 percent (185 of 232 inspections) in 2016, as shown in figure 26. A sector-to-subsector comparison of inspection ratings is provided in figure 27. Compliance to requirements in the radiation protection SCA has been decreasing since 2014. The trend is driven primarily by lower compliance in the nuclear medicine subsector. The most common non-compliance for this SCA amongst medical sector licensees was not performing thyroid monitoring as required.
.png/object)
Description
| Inspection ratings | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Fully satisfactory or satisfactory | 152 | 234 | 192 | 212 | 185 |
| Below expectations | 51 | 53 | 16 | 34 | 46 |
| Unacceptable | 4 | 0 | 1 | 0 | 0 |
.png/object)
Description
| Inspection ratings | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Nuclear medicine | 127 | 185 | 144 | 174 | 143 |
| Radiation therapy | 4 | 21 | 21 | 15 | 24 |
| Veterinary nuclear medicine | 2 | 7 | 8 | 5 | 6 |
| Medical sector | 152 | 238 | 192 | 212 | 185 |
Note: The number of inspections shown in the medical sector row is the aggregate for the entire sector, including subsectors not highlighted in this report.
6.3.5 Security
The compliance rating for the security SCA for licensees in the medical sector was 85.6 percent (190 of 222 inspections) in 2016, as shown in figure 28. All non-compliances were corrected within a short timeframe of being cited.
.png/object)
Description
| Inspection ratings | 2014 | 2015 | 2016 |
|---|---|---|---|
| Fully satisfactory or satisfactory | 181 | 219 | 190 |
| Below expectations | 7 | 4 | 32 |
| Unacceptable | 0 | 0 | 0 |
7 Industrial sector
Licensees in the industrial sector use nuclear substances either in industrial facilities or as part of fieldwork or construction. In 2016, this sector accounted for 1,308 CNSC licences and 43,072 total workers. Of these workers, 12,055 were designated as nuclear energy workers (NEWs).
The results of CNSC staff evaluation of the regulatory performance of all industrial sector licensees inspected in 2016 are included in the overall results. The following four subsectors are highlighted in further detail:
- portable gauge
- fixed gauge
- industrial radiography
- oil well logging
7.1 Sector overview
Typical applications of nuclear substances in the industrial sector include the measurement of physical parameters such as density, moisture content and geological composition in civil engineering. They are also used for level and flow rate in industrial facilities (such as those that support oil and gas exploration, mining and manufacturing). These nuclear substances are found in radiation devices such as fixed nuclear gauges which monitor production processes in many industries, and portable nuclear gauges which are often used to measure moisture and density in soil, and the compaction of asphalt in road construction.
In industrial radiography, nuclear substances are traditionally used in exposure devices for the non-destructive examination of materials. Persons operating these devices, or supervising trainees in the operation of such devices, must be certified by the CNSC. Exposure devices that are used for industrial radiography, as shown in figure 29, are engineered and operated using multiple safety barriers to reduce the potential for accidental occupational exposure. One example is dense material, such as depleted uranium, which shields users against the intense radioactivity of the source contained inside the device.
Industrial applications of nuclear substances are as varied as the processes to which they are applied. Specific radioisotopes are chosen based on the type of radiation they emit, the intensity of their radiation and the intended application. For example, the nuclear substance chosen for industrial radiography depends on the size and density of the material to be imaged. Cobalt-60, with its high-energy gamma radiation, is used for large structures and dense materials such as structural concrete. When the material does not require the penetrating power of cobalt-60, other nuclear substances, such as iridium-192 or selenium-75, are used instead. More recently, a small number of licensees are turning to linear accelerators for radiography imaging. This equipment facilitates analysis of thicker materials than is possible using more traditional methods. Moreover, high-energy CT machines are being used to create three-dimensional images of the interior of materials such as logs and engineered wood products.
Cesium-137, another gamma emitter, is most commonly used in portable and fixed gauges to measure density. In other industrial uses, such as measuring moisture content, portable gauges most commonly use neutron-emitting nuclear substances such as americium-241/beryllium.
.png/object)
7.2 Summary of safety assessment
Based on their evaluation and verification of licensee performance, CNSC staff concluded that the safety performance of the industrial sector was satisfactory in 2016.
Doses received by NEWs in this sector remained low, with the majority of workers receiving doses below 1 millisievert (mSv). No NEW received a dose in excess of the annual regulatory limits.
Of all the inspected licensees in 2016, the majority of them were found to be compliant in the four SCAs covered in this report:
- 98.4 percent were compliant in management system
- 86.4 percent were compliant in operating performance
- 84.4 percent were compliant in radiation protection
- 95.1 percent were compliant in security
In cases where non-compliances were noted, licensees took appropriate corrective actions, satisfactory to CNSC staff, to address the non-compliances.
The CNSC took 18 escalated enforcement actions (12 orders and six administrative monetary penalties) against licensees in the industrial sector in 2016. Further details are provided in section 7.3.6.
7.3 Safety performance measures
7.3.1 Doses to workers
NEWs in the industrial radiography subsector continued to receive higher doses than workers in other industrial subsectors, as shown in figure 30. This is a result of working in close proximity to exposure devices containing high activity sealed sources. figure 31 shows the doses to NEWs in the industrial radiography subsector over the period of 2012 to 2016. Doses to workers on the higher end of the spectrum (i.e., between 20 and 50 mSv) continue to decrease.
.png/object)
Description
| Industrial sector | ≤ 0.5 | > 0.5 and ≤ 1 mSv | > 1 and ≤ 5 mSv | > 5 and ≤ 20 mSv | > 20 and ≤ 50 mSv | > 50 mSv |
|---|---|---|---|---|---|---|
| Portable gauge | 2,355 | 1,479 | 931 | 17 | – | – |
| Fixed gauge | 66 | – | – | – | – | – |
| Industrial radiography | 1,415 | 380 | 873 | 314 | 1 | – |
| Oil well logging | 1,103 | 48 | 40 | 6 | – | – |
| Industrial sector | 5,123 | 1,922 | 1,851 | 337 | 1 | – |
Note: The total number of NEWs shown in the industrial sector row is the aggregate for the entire sector, including subsectors not highlighted in this report.
.png/object)
Description
| Year | ≤ 0.5 | > 0.5 and ≤ 1 mSv | > 1 and ≤ 5 mSv | > 5 and ≤ 20 mSv | > 20 and ≤ 50 mSv | > 50 mSv |
|---|---|---|---|---|---|---|
| 2012 | 227 | 38 | 147 | 49 | – | – |
| 2013 | 1,026 | 346 | 878 | 409 | 11 | – |
| 2014 | 1,257 | 337 | 885 | 360 | 6 | – |
| 2015 | 1,151 | 365 | 924 | 396 | 3 | – |
| 2016 | 1,415 | 380 | 873 | 314 | 1 | – |
7.3.2 Management system
The compliance rating for management system in the industrial sector was 98.3 percent (901 of 917 inspections) in 2016, as shown in figure 32. Two licensees received unacceptable ratings for this SCA. Both received an order from a CNSC inspector. Details of the circumstances can be found in section 7.3.6 and appendix C.
Among industrial sector licensees, the main non-compliances in this SCA involved licensees not notifying the CNSC of locations where they conducted licensed activities for more than 90 days, not having the required records at temporary locations used for more than 90 days, and failing to notify the CNSC of changes to their applicant authority or radiation safety officers.
.png/object)
Description
| Sector | 2015 | 2016 |
|---|---|---|
| Portable gauge | 384 | 436 |
| Fixed gauge | 163 | 204 |
| Industrial radiography | 157 | 194 |
| Oil well logging | 49 | 48 |
| Industrial sector | 837 | 901 |
Note: The number of inspections shown in the industrial sector row is the aggregate for the entire industrial sector, including subsectors not highlighted in this report.
7.3.3 Operating performance
The compliance rating for operating performance in the industrial sector was 86.4 percent (792 of 917 inspections) in 2016, as shown in figure 33. Five licensees received unacceptable ratings for operational performance. CNSC inspectors issued orders in all five cases. The circumstances of each are listed in section 7.3.6 and in appendix C. A sector-to-subsector comparison of inspection ratings is provided in figure 34. After a year with high compliance ratings in 2015, the fixed gauge subsector’s rating in this SCA dropped back to the level observed in 2014. Conversely, both the industrial radiography and oil well logging subsectors improved their performance in this SCA compared with 2015. For the industrial radiography subsector, this is a continuation of steadily improving compliance, and for the oil well logging subsector, the improvement seen in 2016 is a reversal of a downward trend from 2013 to 2015.
The most common types of non-compliances were workers failing to follow the licensee’s procedures and using the safety equipment provided to them, licensees failing to follow the procedures in their radiation safety manuals, and licensees failing to keep required training records.
.png/object)
Description
| Inspection ratings | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Fully satisfactory or satisfactory | 781 | 839 | 839 | 785 | 792 |
| Below expectations | 98 | 115 | 115 | 78 | 120 |
| Unacceptable | 5 | 4 | 1 | 2 | 5 |
.png/object)
Description
| Sector | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Portable gauge | 331 | 357 | 399 | 356 | 382 |
| Fixed gauge | 131 | 160 | 167 | 153 | 157 |
| Industrial radiography | 244 | 204 | 203 | 175 | 187 |
| Oil well logging | 57 | 60 | 53 | 39 | 43 |
| Industrial sector | 781 | 839 | 839 | 785 | 792 |
Note: The number of inspections shown in the industrial sector row is the aggregate for the entire industrial sector, including subsectors not highlighted in this report.
7.3.4 Radiation protection
The compliance rating for the radiation protection SCA for licensees in the industrial sector was 84.4 percent (773 of 916 inspections) in 2016, as shown in Figure 35. A sector-to-subsector comparison of inspection ratings is provided in figure 36. The oil well logging and portable gauge subsectors have continued downward trends in compliance in the radiation protection SCA.
Four licensees from the industrial sector received unacceptable ratings. CNSC inspectors issued orders to each of these licensees. The circumstances of each situation can be found in section 7.3.6 and appendix C.
The most common non-compliances involved licensees not having available a calibrated survey meters, inadequate implementation of radiation protection programs that keep doses to workers and the public as low as reasonably achievable, and failing to post radiation warning signs.
.png/object)
Description
| Inspection ratings | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Fully satisfactory or satisfactory | 771 | 833 | 842 | 768 | 773 |
| Below expectations | 123 | 118 | 111 | 94 | 139 |
| Unacceptable | 6 | 4 | 3 | 0 | 4 |
.png/object)
Description
| Sector | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Portable gauge | 327 | 367 | 400 | 355 | 372 |
| Fixed gauge | 139 | 150 | 179 | 136 | 159 |
| Industrial radiography | 230 | 201 | 192 | 173 | 182 |
| Oil well logging | 62 | 63 | 55 | 43 | 30 |
| Industrial sector | 771 | 833 | 842 | 768 | 773 |
Note: The number of inspections shown in the industrial sector row is the aggregate for the entire industrial sector, including subsectors not highlighted in this report.
7.3.5 Security
The compliance rating for security SCA for licensees in the industrial sector was 95.1 percent (830 of 873 inspections) in 2016, as shown in figure 37. One licensee received an unacceptable rating in this SCA, and a CNSC inspector issued an order to the licensee. As a result, the licensee implemented corrective measures to the satisfaction of the CNSC and the order has been closed.
.png/object)
Description
| Inspection ratings | 2014 | 2015 | 2016 |
|---|---|---|---|
| Fully satisfactory or satisfactory | 875 | 780 | 830 |
| Below expectations | 56 | 46 | 42 |
| Unacceptable | 0 | 2 | 1 |
7.3.6 Enforcement actions
The CNSC took 18 escalated enforcement actions against licensees in the industrial sector in 2016. These consisted of 12 orders and six administrative monetary penalties (AMPs). The number of enforcement actions taken against licensees in this sector has continued to rise since 2014. CNSC staff monitor the number of enforcement actions closely and take necessary measures to correct negative trends.
The CNSC issued seven orders against licensees in the portable gauge subsector, two in the industrial radiography subsector, two in the fixed gauge subsector and one in the oil well logging subsector. The distribution of orders by industrial subsectors from 2012 to 2016 is shown in figure 38.
The CNSC issued four AMPs to portable gauge licensees. Two were issued in conjunction with orders described above. One was issued in conjunction with an order issued in 2015. The fourth was issued as a result of the CNSC’s investigation of an event reported by the licensee.
In 2016, the CNSC issued enforcement actions to three fixed gauge licensees that performed vessel entry procedures without following the safety requirements prescribed by the CNSC in their licence conditions. One licensee received an order (listed above) and an AMP in 2016, the second received an order in 2016 (also listed above) and an AMP in 2017, and the third licensee received an AMP in 2016. The first two situations were identified during CNSC inspections, while the third case was brought to the attention of CNSC staff through mandatory event reporting by the licensee. In early 2017, in response to the non-compliances with requirements for entry into a vessel for which radiation devices are used, CNSC staff developed and distributed an information bulletin highlighting the requirements for safe vessel and hopper entry.
All licensees to which orders were issued complied with the terms and conditions of the orders and implemented corrective measures to the satisfaction of CNSC staff. The licensees that were issued AMPs have paid their penalty amounts.
.png/object)
Description
| Industrial sector | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Portable gauge | 9 | 14 | 3 | 4 | 11 |
| Fixed gauge | 2 | 1 | 2 | 3 | 4 |
| Industrial radiography | 5 | 5 | 4 | 3 | 2 |
| Other | – | 2 | 1 | 3 | 1 |
| Industrial sector | 16 | 22 | 10 | 13 | 18 |
Details of all enforcement actions issued in 2016 are provided in appendix C. Further information on regulatory actions, including escalated enforcement actions, taken by the CNSC is available on the CNSC website.
8 Academic and research sector
Licensed activities in the academic and research sector are conducted in universities, colleges and research laboratories. In 2016, this sector accounted for 208 licences and 7,240 total workers, of which 2,670 were designated as nuclear energy workers (NEWs).
Safety performance results are provided for all licensees included in the academic and research sector, with the laboratory studies and consolidated uses of nuclear substances subsectors highlighted in further detail.
.png/object)
8.1 Sector overview
This sector focuses mainly on biological and biomedical research that primarily uses open (unsealed) nuclear substances, as shown in figure 39. The sector also uses sealed sources, radiation devices and accelerators for teaching as well as for pure and applied research.
CNSC laboratory
As part of its regulatory functions, the CNSC conducts certain activities regulated under the Nuclear Safety and Control Act (NSCA). To ensure oversight transparency, CNSC management has separated the organization’s work as a licensee (which resides within the Technical Support Branch) from its work as a regulator (under the responsibility of the Regulatory Operations Branch).
The CNSC laboratory provides calibration services and analytical services for CNSC staff, including CNSC inspectors. To provide these services, the CNSC holds two licences: one for its gamma calibration irradiator located at its laboratory in Ottawa, and a second for consolidated uses of nuclear substances that covers all other activities conducted by the CNSC at its laboratory or elsewhere in Canada. Both licences were issued in accordance with the NSCA and are regulated using the same licensing and compliance verification processes that would apply to other, similar licensees.
In this report, the CNSC laboratory is included in the laboratory studies and consolidated use of nuclear substances subsector. Its specific performance results are provided to demonstrate that the CNSC, as both regulator and licensee, is reporting on its licensed activities in a transparent manner.
Doses received by NEWs working at the CNSC laboratory remained very low, with all workers receiving doses below 1 mSv.
In June 2016, CNSC staff conducted an unannounced inspection of an internal permit holder of the CNSC laboratory’s consolidated uses of nuclear substances licence. No items of non-compliance were identified during the inspection.
8.2 Summary of safety assessment
The academic and research sector continued to show satisfactory safety performance in 2016.
Doses received by NEWs in this sector remained very low, with the majority of workers receiving doses below 1 mSv.
Of all the inspected licensees in 2016, the majority were found to be compliant in the four safety and control areas (SCAs) covered in this report:
- 97.3 percent were compliant in management system
- 91.4 percent were compliant in operating performance
- 91 percent were compliant in radiation protection
- 95.9 percent were compliant in security
In cases where non-compliances were noted during inspections, licensees took appropriate corrective actions, satisfactory to CNSC staff, to address the non-compliances.
No enforcement actions were taken against licensees in the academic and research sector in 2016.
8.3 Sector performance measures
8.3.1 Doses to workers
Doses received by NEWs in this sector remained very low. The majority of workers received doses below 1 mSv, as shown in figure 40.
.png/object)
Description
| Sector | ≤ 0.5 | > 0.5 and ≤ 1 mSv | > 1 and ≤ 5 mSv | > 5 and ≤ 20 mSv | > 20 and ≤ 50 mSv | > 50 mSv |
|---|---|---|---|---|---|---|
| Laboratory studies and consolidated use of nuclear substances | 2,310 | 41 | 38 | 3 | – | – |
| Academic and research sector | 2,575 | 49 | 41 | 5 | – | – |
Note: The total number of NEWs shown in the academic and research sector row is the aggregate for the entire sector, including subsectors not highlighted in this report.
8.3.2 Management system
In 2016, the compliance rating for management system in the academic and research sector was 97.3 percent (73 of 75 inspections), as shown in figure 41. No licensees received unacceptable ratings in this SCA. A sector-to-subsector comparison is shown in figure 42.
.png/object)
Description
| Inspection rating | 2015 | 2016 |
|---|---|---|
| Fully satisfactory or satisfactory | 678 | 73 |
| Below expectations | 4 | 2 |
| Unacceptable | 0 | 0 |
.png/object)
Description
| Sector | 2015 | 2016 |
|---|---|---|
| Laboratory studies and consolidated use of nuclear substances | 58 | 69 |
| Academic and research sector | 67 | 73 |
Note: The number of inspections shown in the academic and research row is the aggregate for the entire sector, including subsectors not highlighted in this report.
8.3.3 Operating performance
The overall compliance rating for operating performance in the academic and research sector was 91.4 percent (74 of 81 inspections) in 2016, as shown in figure 43. This is a significant improvement in performance over 2015, when only 77.4 percent of inspected licensees were compliant to requirements in this SCA. A sector-to-subsector comparison for operating performance ratings is provided in figure 44. The laboratory studies and consolidated uses of nuclear substances subsector is the main driver of the rating in this SCA. The main non-compliance involved not following procedures.
In 2014, the CNSC inspection program for the laboratory studies and consolidated uses of nuclear substances subsector was revised based on the positive safety performance ratings and the low-risk level associated with these licensed activities. The frequency of CNSC inspections was changed from annually to every two years, which is reflected in the decrease in the number of inspections conducted since 2014 for this subsector.
2012–16
.png/object)
Description
| Inspection rating | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Fully satisfactory or satisfactory | 223 | 178 | 118 | 60 | 74 |
| Below expectations | 41 | 18 | 17 | 17 | 7 |
| Unacceptable | 0 | 0 | 0 | 0 | 0 |
.png/object)
Description
| Sector | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Laboratory studies and consolidated uses | 217 | 172 | 108 | 47 | 75 |
| Academic and research sector | 223 | 178 | 118 | 60 | 81 |
Note: The number of inspections shown in the academic and research row is the aggregate for the entire sector, including subsectors not highlighted in this report.
8.3.4 Radiation protection
The overall compliance rating for radiation protection in the academic and research sector was 91 percent (71 of 78 inspections) in 2016, as shown in figure 45. A sector-to-subsector comparison for radiation protection ratings is provided in figure 46.
.png/object)
Description
| Inspection ratings | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Fully satisfactory or satisfactory | 213 | 174 | 117 | 65 | 71 |
| Below expectations | 50 | 21 | 17 | 7 | 7 |
| Unacceptable | 0 | 0 | 0 | 0 | 0 |
.png/object)
Description
| Sector | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Laboratory studies and consolidated uses | 207 | 167 | 106 | 53 | 66 |
| Academic and research sector | 213 | 174 | 117 | 65 | 71 |
Note: The number of inspections shown in the academic and research row is the aggregate for the entire sector, including subsectors not highlighted in this report.
8.3.5 Security
The compliance rating for security SCA for licensees in the academic and research sector was 95.9 percent (70 of 73 inspections) in 2016, as shown in figure 47. This is consistent with the sector’s performance over the last two years.
.png/object)
Description
| Inspection ratings | 2014 | 2015 | 2016 |
|---|---|---|---|
| Fully satisfactory or satisfactory | 120 | 64 | 70 |
| Below expectations | 3 | 6 | 3 |
| Unacceptable | 0 | 0 | 0 |
9 Commercial sector
The commercial sector encompasses a number of licensed activities related to the production, processing, storage and distribution of nuclear substances, the calibration of radiation detection instruments, as well as the servicing of radiation devices and Class II prescribed equipment as a commercial enterprise. In 2016, this sector accounted for 247 CNSC licences and 1,899 total workers, including 1,378 designated as nuclear energy workers (NEWs).
Safety performance results are provided for all licensees included in the commercial sector, with the following five subsectors highlighted in further detail:
- isotope production accelerators
- processing of nuclear substances
- distribution of nuclear substances
- servicing of radiation devices and prescribed equipment
- calibration of radiation devices and prescribed equipment
Figure 48 shows the internal components of a partially assembled cyclotron used for the production of radioisotopes.

9.1 Sector overview
The commercial sector encompasses a number of licensed activities related to the production, processing, storage and distribution of nuclear substances, and the calibration and servicing of radiation devices for commercial gain.
Isotope-production cyclotrons can produce a range of different radioisotopes that are widely used in the diagnosis, management and treatment of disease. Most licensees in the processing of nuclear substances subsector process isotopes to provide products and services used for the prevention, diagnosis and treatment of disease. Distributors of radiation devices and nuclear substances are the link between the manufacturer and the end user. In some cases (for example, smoke detectors), end users are not required to hold licences for devices; however, those companies that distribute such products in Canada are. A licence is required to calibrate radiation detection instruments such as radiation survey meters. Licensees with calibration licences use nuclear substances and radiation devices to determine the response of radiation detection instruments. Installation, repair and non-routine maintenance of radiation devices and prescribed equipment located in Canada requires a servicing licence issued by the CNSC, even if the licensee’s headquarters is located outside Canada.
9.2 Summary of safety assessment
The commercial sector continued to show good safety performance in 2016.
Doses received by NEWs in this sector remained low. The majority of workers received doses below 1 millisievert (mSv).
Of all the inspected licensees in 2016, the majority were found to be compliant in the four SCAs covered in this report:
- 96.4 percent were compliant in management system
- 92.0 percent were compliant in operating performance
- 91.8 percent were compliant in radiation protection
- 98.5 percent were compliant in security
In cases where non-compliances were noted during the inspection, licensees took appropriate corrective actions, satisfactory to CNSC staff, to address the non-compliances.
The CNSC took escalated enforcement actions against two licensees in the commercial sector.
- The CNSC issued an order and an administrative monetary penalty (AMP) to a licensee that offers calibration services. The order was issued during an inspection when the CNSC inspector determined that the licensee had serious gaps in the implementation of its radiation safety program and worker training. The AMP was issued following an assessment by the CNSC during which inspectors discovered that the licensee had presented for transport, and subsequently transported, nuclear substances in packages that did not meet the applicable regulatory requirements. CNSC staff reviewed the corrective measures implemented by the licensee and found them to be satisfactory. The licensee has paid the AMP.
- The CNSC also issued an order to a licensee that produces radiopharmaceuticals for servicing prescribed equipment without a licence. Additional details about the enforcement actions are found in appendix C.
Additional details about the enforcement actions are found in appendix C.
9.3 Safety performance measures
9.3.1 Doses to workers
NEWs in the isotope production accelerators and processing of nuclear substances subsectors continued to receive higher doses than workers in other commercial subsectors, as shown in figure 49. This is due to their manual handling of nuclear substances and activated cyclotron components. The vast majority of NEWs in these subsectors received doses below 5 mSv in 2016.
Annual effective doses for NEWs in the isotope production accelerators subsector from 2012 to 2016 are shown in figure 50. Annual effective doses for NEWs in the processing of nuclear substances subsector from 2012 to 2016 are shown in figure 51.
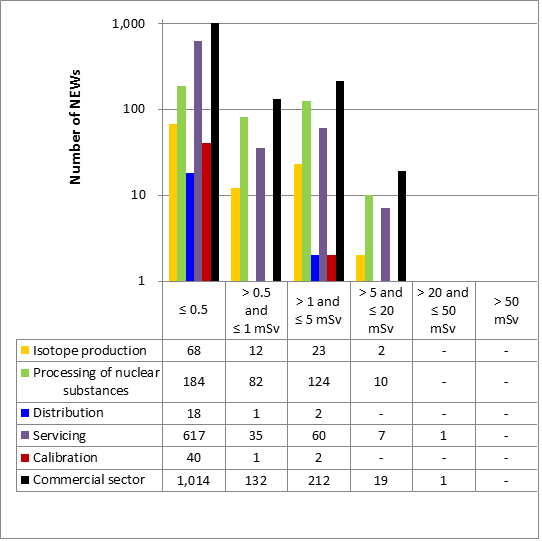
Description
| Sector | ≤ 0.5 | > 0.5 and ≤ 1 mSv | > 1 and ≤ 5 mSv | > 5 and ≤ 20 mSv | > 20 and ≤ 50 mSv | > 50 mSv |
|---|---|---|---|---|---|---|
| Isotope production | 68 | 12 | 23 | 2 | – | – |
| Processing of nuclear substances | 184 | 82 | 124 | 10 | – | – |
| Distribution | 18 | 1 | 2 | – | – | – |
| Servicing | 617 | 35 | 60 | 7 | 1 | – |
| Calibration | 40 | 1 | 2 | – | – | – |
| Commercial sector | 1,014 | 132 | 212 | 19 | 1 | – |
Note: The total number of NEWs shown in the commercial sector is the aggregate for the entire sector, including subsectors not highlighted in this report.
2012–16
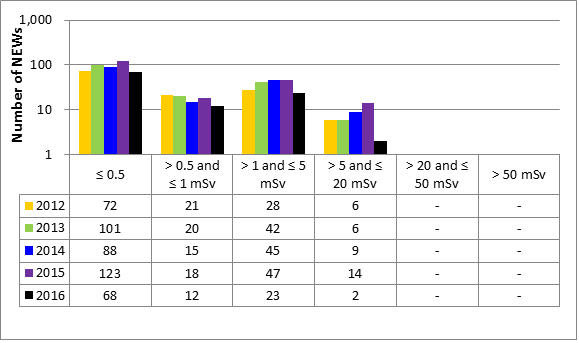
Description
| Year | ≤ 0.5 | > 0.5 and ≤ 1 mSv | > 1 and ≤ 5 mSv | > 5 and ≤ 20 mSv | > 20 and ≤ 50 mSv | > 50 mSv |
|---|---|---|---|---|---|---|
| 2012 | 72 | 21 | 28 | 6 | – | – |
| 2013 | 101 | 20 | 42 | 6 | – | – |
| 2014 | 88 | 15 | 45 | 9 | – | – |
| 2015 | 123 | 18 | 47 | 14 | – | – |
| 2016 | 68 | 12 | 23 | 2 | – | – |
2012–16
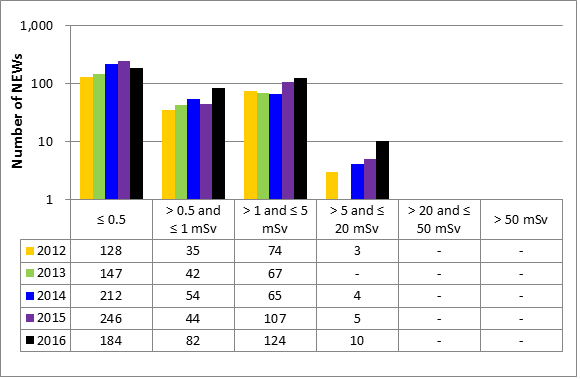
Description
| Year | ≤ 0.5 | > 0.5 and ≤ 1 mSv | > 1 and ≤ 5 mSv | > 5 and ≤ 20 mSv | > 20 and ≤ 50 mSv | >50 mSv |
|---|---|---|---|---|---|---|
| 2012 | 128 | 35 | 74 | 3 | – | – |
| 2013 | 147 | 42 | 67 | – | – | – |
| 2014 | 212 | 54 | 65 | 4 | – | – |
| 2015 | 246 | 44 | 107 | 5 | – | – |
| 2016 | 184 | 82 | 124 | 10 | – | – |
9.3.2 Management system
Commercial sector licensees had an overall compliance rating in the management system SCA of 96.4 percent (80 of 83 inspections). All three instances of non-compliance were due to licensees conducting activities outside the scope of their licences. An order and an AMP were issued to one licensee as a result of its non-compliance to this requirement.
The compliance ratings for the sector as a whole are shown in figure 52. Figure 53 shows the breakdown of compliant inspections by subsector. The calibration subsector showed a decrease in compliance to requirements in the management system SCA compared to 2015. Because there were only seven inspections of calibration licensees, and this is only the second year the CNSC has presented data on management systems, it is too early to determine whether it is a trend.
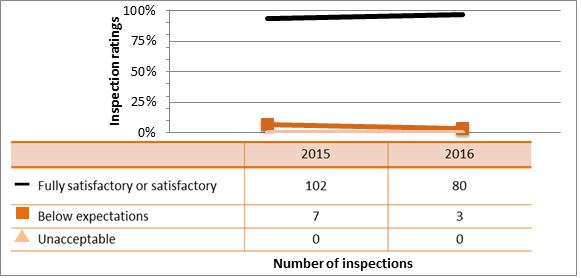
Description
| Inspection ratings | 2015 | 2016 |
|---|---|---|
| Fully satisfactory or satisfactory | 102 | 80 |
| Below expectations | 7 | 3 |
| Unacceptable | 0 | 0 |
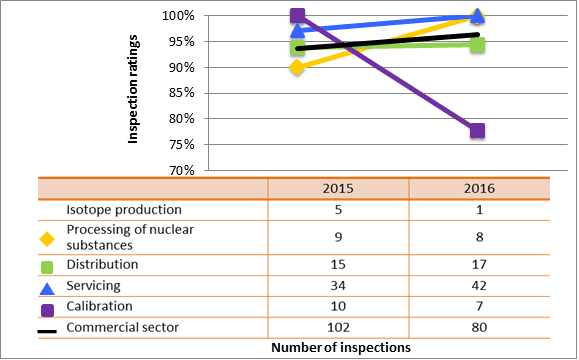
Description
| Commercial subsector | 2015 | 2016 |
|---|---|---|
| Isotope production | 5 | 1 |
| Processing of nuclear substances | 9 | 8 |
| Distribution | 15 | 17 |
| Servicing | 34 | 42 |
| Calibration | 10 | 7 |
| Commercial sector | 102 | 80 |
Note: The number of inspections shown in the commercial sector row is the aggregate for the entire commercial sector, including subsectors not highlighted in this report. The trend line was not provided for the isotope production accelerators subsector due to the low number of inspections conducted.
9.3.3 Operating performance
The overall compliance rating in 2016 for operating performance in the commercial sector was 92 percent (80 of 87 inspections), as shown in figure 54. A sector-to-subsector comparison for operating performance ratings is provided in figure 55.
The most common non-compliances involved workers not following licensee procedures or not using the provided safety equipment, and licensees not keeping records for servicing work they performed for others.
The processing of nuclear substance subsector has showed a downward trend in compliance ratings since 2013.
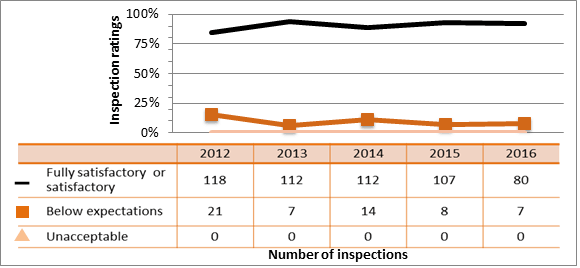
Description
| Inspection ratings | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Fully satisfactory or satisfactory | 118 | 112 | 112 | 107 | 80 |
| Below expectations | 21 | 7 | 14 | 8 | 7 |
| Unacceptable | 0 | 0 | 0 | 0 | 0 |
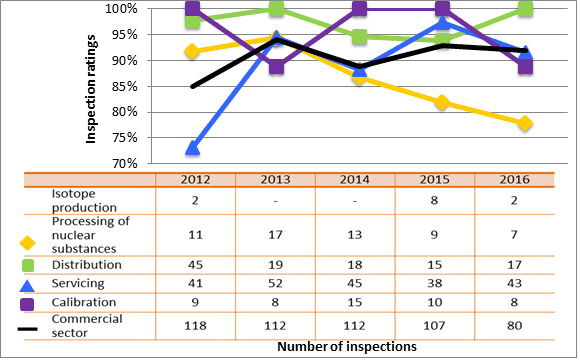
Description
| Commercial subsector | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Isotope production | 2 | – | – | 8 | 2 |
| Processing of nuclear substances | 11 | 17 | 13 | 9 | 7 |
| Distribution | 45 | 19 | 18 | 15 | 17 |
| Servicing | 41 | 52 | 45 | 38 | 43 |
| Calibration | 9 | 8 | 15 | 10 | 8 |
| Commercial sector | 118 | 112 | 112 | 107 | 80 |
Note: The number of inspections shown in the commercial sector row is the aggregate for the entire commercial sector, including subsectors not highlighted in this report. The trend line was not provided for the isotope production accelerators subsector due to low number of inspections conducted.
9.3.4 Radiation protection
The overall compliance rating for radiation protection in the commercial sector was 91.9 percent (79 of 86 inspections) in 2016, as shown in figure 56. A sector-to-subsector comparison for radiation protection ratings is provided in figure 57.
The most common non-compliance was licensees failing to implement radiation protection programs that keep doses to workers and the public ALARA.
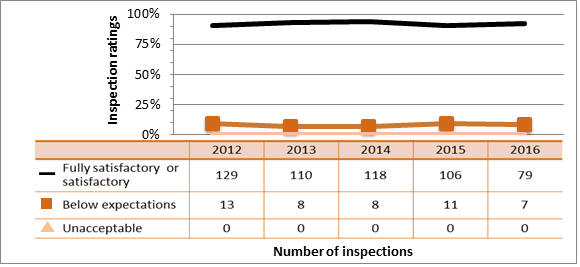
Description
| Inspection ratings | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Fully satisfactory or satisfactory | 129 | 110 | 118 | 106 | 79 |
| Below expectations | 13 | 8 | 8 | 11 | 7 |
| Unacceptable | 0 | 0 | 0 | 0 | 0 |
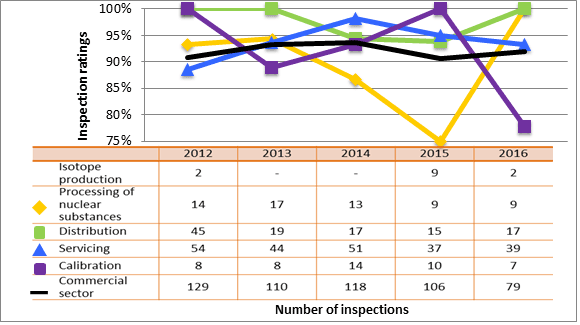
Description
| Commercial subsectors | 2012 | 2013 | 2014 | 2015 | 2016 |
|---|---|---|---|---|---|
| Isotope production | 2 | – | – | 9 | 2 |
| Processing of nuclear substances | 14 | 17 | 13 | 9 | 9 |
| Distribution | 45 | 19 | 17 | 15 | 17 |
| Servicing | 54 | 44 | 51 | 37 | 39 |
| Calibration | 8 | 8 | 14 | 10 | 7 |
| Commercial | 129 | 110 | 118 | 106 | 79 |
Note: The number of inspections shown in the commercial sector row is the aggregate for the entire commercial sector, including subsectors not highlighted in this report. The trend line was not provided for the isotope production accelerators subsector due to low number of inspections conducted.
9.3.5 Security
The compliance rating for security SCA for licensees in the commercial sector was 98.5 percent (65 of 66 inspections) in 2016, as shown in figure 58.
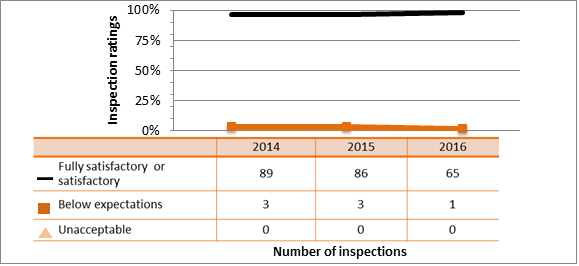
Description
| Inspection ratings | 2014 | 2015 | 2016 |
|---|---|---|---|
| Fully satisfactory or satisfactory | 89 | 86 | 65 |
| Below expectations | 3 | 3 | 1 |
| Unacceptable | 0 | 0 | 0 |
10 Conclusion
CNSC staff continued their ongoing regulatory oversight of licensees in the medical, industrial, academic and research, and commercial sectors. Staff conducted compliance verification activities consisting of field inspections, desktop reviews and technical assessments of licensee activities, and concluded that the use of nuclear substances in Canada is safe. The evaluations of findings for the safety and control areas (SCAs) covered in this report show that, overall, licensees made adequate provisions for the protection of the health, safety and security of persons and the environment from the use of nuclear substances, and took the measures required to implement Canada’s international obligations.
Compliance verification
In 2016, CNSC staff conducted 1,452 inspections to verify compliance with CNSC regulatory requirements across all sectors, including 228 security inspections related to the implementation of REGDOC-2.12.3, Security of Nuclear Substances: Sealed Sources, and three inspections related to the export of high-risk sealed sources. Of inspected licensees, the majority were found to be compliant in the four SCAs covered in this report:
- 97.5 percent were compliant in management system
- 87.4 percent were compliant in operating performance
- 84.6 percent were compliant in radiation protection
- 93.6 percent were compliant in security
Those licensees failing to meet requirements took appropriate corrective measures to address non-compliances found during inspections. CNSC staff systematically tracked all non-compliances until licensees took the appropriate corrective measures to address them. All corrective measures put in place by licensees were reviewed by CNSC staff and found to be satisfactory.
Effective doses to workers
Doses to workers remained very low in 2016, consistent with previous years. One of the 22,606 nuclear energy workers (NEWs) received an equivalent dose above the CNSC regulatory dose limit of 500 millisieverts (mSv) for the hands and feet. Appropriate corrective actions were taken by the licensee in response to this event, which CNSC staff reported to the Commission in December 2016. None of the workers designated as NEWs exceeded the one-year effective dose limit of 50 mSv.
Enforcement actions
In 2016, the CNSC took escalated enforcement actions in 22 instances. It issued 14 orders and eight administrative monetary penalties to ensure that the health and safety of workers, the Canadian public and the environment were being adequately protected. Most of the enforcement actions were taken against licensees in the industrial sector, consistent with trends from previous years. All licensees to whom orders were issued have implemented corrective measures, which were reviewed by CNSC staff and found to be satisfactory. All eight administrative monetary penalties (AMPs) issued in 2016 have been paid.
Reported events
Licensees reported 139 events to the CNSC that are covered in this report – all of which were assessed by CNSC staff. Of the total number of events reported, 136 were categorized as level 0 (no safety significance) on the International Nuclear and Radiation Events Scale. A further two events were ranked as level 1 (anomaly) due to the quantity of nuclear substances involved and the type of event reported (i.e., the loss of nuclear substances). The remaining event – ranked at level 2 (incident) – resulted in a NEW receiving a dose to the hands dose above the regulatory limit.
There were no releases of nuclear substances to the environment that had an adverse radiological impact or that resulted in a person receiving a dose in excess of the regulatory limit for members of the public.
One member of the public received a dose in excess of the annual regulatory dose limit of 1 mSv after taking a ride from a ride-sharing service in a vehicle in which the driver was also transporting packages containing nuclear substances. The event was reported to the Commission in December 2016. An AMP was issued to the driver in 2017.
For all reported events, licensees implemented appropriate response measures to mitigate the impacts of the events and to limit radiation exposure to workers and the public. These measures were reviewed by CNSC staff and found to be satisfactory.
Regulatory focus in 2017
In 2017, the CNSC will continue to focus on effective regulatory oversight and continuous improvement, with greater emphasis on:
- using Type I inspections for compliance verification of large licensees
- clarifying expectations for reportable events with the development of regulatory document REGDOC-3.1.2 Part II – Reporting Requirements for Nuclear Substances and Radiation Devices
- enhancing oversight of radiation safety officers across all sectors
- preparing licensees with Category 3, 4 and 5 sealed sources to be compliant with the expectations of REGDOC-2.12.3, Security of Nuclear Substances: Sealed Sources which comes into effect May 31, 2018
- increased focus on compliance verification of fixed gauge licensees with the licence condition for vessel or hopper entry
- implementing regulatory oversight programs for new devices and technologies, particularly in the healthcare sector
- an increased number of applications for certification of prescribed equipment and radiation devices due to the expiry of many existing certificates
Conclusion
The use of nuclear substances in Canada is safe. Adequate provisions for the protection of the health, safety and security of persons and the environment from the use of nuclear substances are in place.
Appendix A: Radiation exposure
Non-occupational exposure to radiation can occur in many situations. For example, a person may be exposed to radiation during an airplane flight or by undergoing a medical procedure such as a chest X-ray. Natural background radiation contributes to radiation exposure received by all persons living on earth. The average annual dose from natural background radiation is approximately 1.8 millisieverts (mSv) in Canada and 2.4 mSv worldwide. Among major Canadian cities, Winnipeg has the highest annual average dose from background radiation at 4.1 mSv.
Figure 59 provides some perspective on these situations as they relate to doses to the public as well as occupational radiation exposures received by workers as a result of both nuclear activities licensed by the CNSC.
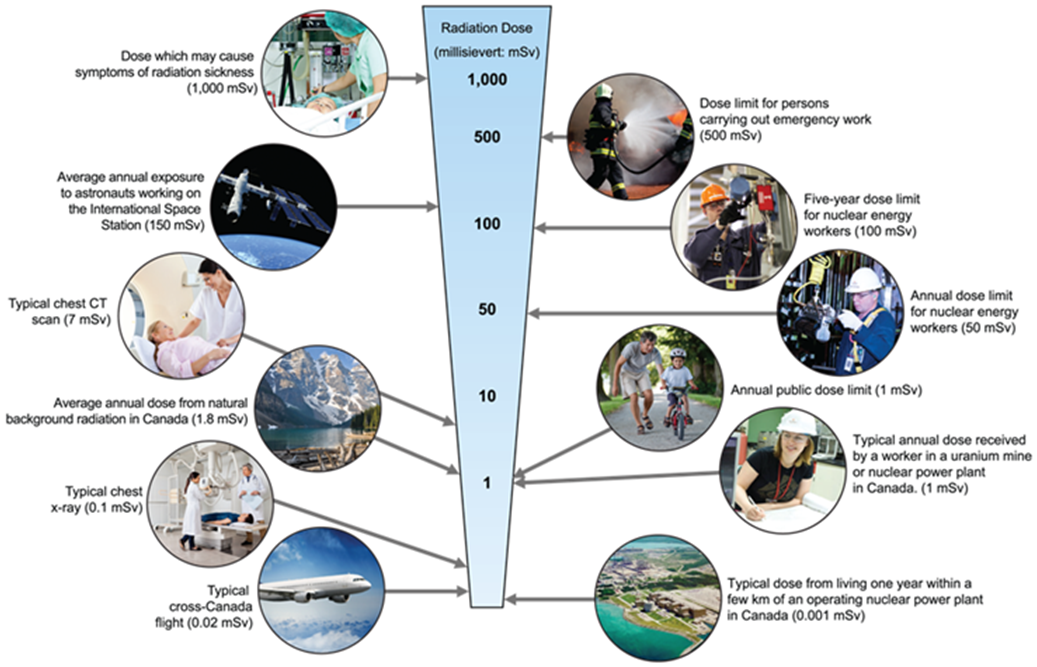
Ascertaining effective dose
In this report, effective dose refers to the dose received by the whole body. Each licensee is required to ascertain the effective dose received by each worker engaged in activities authorized under their CNSC licence. Doses may be ascertained by direct measurement (through monitoring) or by estimation, in accordance with the Radiation Protection Regulations. The Radiation Protection Regulations also stipulate that the licensee must use a licensed dosimetry service for monitoring every nuclear energy worker who has a reasonable probability of receiving an effective dose of greater than 5 mSv per year. However, regardless of the potential for occupational exposure, licensees conducting licensed activities in certain industries, such as industrial radiography, are always required to use a licensed dosimetry service provider to ascertain doses for the nuclear energy workers they employ (under subsection 30(3) of the Nuclear Substances and Radiation Devices Regulations).
When a dose limit is exceeded
In a situation where a worker may have exceeded a regulatory dose limit, licensees are required to remove the worker from any activities that may add to his or her dose, investigate the cause of the exposure, take action to prevent a recurrence, and report to the CNSC. CNSC staff review the information provided by the licensee following each investigation. Depending on the circumstances, the Commission, or in most cases a designated officer authorized by the Commission, may authorize the worker to return to work according to the process defined in the Radiation Protection Regulations. The return-to-work authorization may specify conditions and prorated dose limits for the remainder of the dosimetry period.
Appendix B: Safety and control area naming conventions
Safety and control areas (SCAs) used in this report reflect the standardized set and naming convention approved for CNSC licensed activities, as shown the left-hand column of Table 5. For historical reasons, a modified naming convention of SCAs is used for the inspections of nuclear substances activities covered in this report (i.e., licensees that use nuclear substances). See the right-hand column of Table 5. In the near future, the CNSC intends to adopt the standardized naming convention of SCAs for all types of licensees that use nuclear substances. It should be noted that not all SCAs are considered for the inspection of nuclear substances activities and facilities.
| Safety and control area | Safety and control area: Inspection reports |
|---|---|
| Management system | Organization and management Quality management |
| Human performance management | Training and qualification |
| Operating performance | Operational procedures |
| Safety analysis | Facility shielding design Facility safety systems |
| Physical design | Facility shielding design Facility safety systems |
| Fitness for service | Entrance and exit monitors Alarms and status machines Fault indicators |
| Radiation protection | Radiation protection |
| Conventional health and safety | Non-radiological health and safety |
| Environmental protection | Environmental protection |
| Emergency management and fire protection | Emergencies and unplanned events Fire protection |
| Waste management | Environmental protection |
| Security | Security |
| Safeguards | International obligations and safeguards |
| Packaging and transport | Packaging and transport |
Appendix C: Enforcement actions issued in 2016
CNSC designated officers issued a total of 22 enforcement actions in the form of 14 orders and eight administrative monetary penalties (AMPs) in 2016. Details of the orders issued are shown in table 6. Details of AMPs are provided in table 7.
| Issue date and location | Licensee (subsector) | Licensee response | Closure date |
|---|---|---|---|
| March 10 (Fort Saskatchewan, Alberta) | PML Inspection Services Ltd. (Industrial radiography) | Ceased all radiography operations until an effective radiation protection program was implemented. Corrected all items of non-compliance to the CNSC’s satisfaction. | March 21 |
| March 11 (Sarnia, Ontario) | Tomlinson Enterprises Ltd. (Industrial radiography | Ceased all radiography operations until all exposure devices were serviced by a qualified individual, and all exposure device operators and trainees received training on the licensee’s procedures for transporting radioactive sources and exposure devices | March 16 |
| March 18 (Merlin, Ontario) | Nuclear Services Canada Inc. (Calibration) | Ceased all activities involving radioactive materials until: all employees received training on the licensee’s radiation safety program, demonstrated compliance with all requirements for the transportation of radioactive materials, and corrected all items of non-compliance to the satisfaction of the CNSC. Conducted worker dose ascertainments and submitted the results to the CNSC | May 2 |
| March 24 (Sarnia, Ontario) | Canadian Tower Scanning Inc. (Oil well logging) | Ceased all activities involving radioactive materials until all employees received training on all aspects of the licensee’s radiation safety program. Corrected all items of non-compliance to the satisfaction of the CNSC. Conducted a complete dose ascertainment for workers handling radioactive sources and submitted the results to the CNSC. | April 20 |
| June 1 (Vernon, British Columbia) | Hoban Equipment Ltd. (Portable gauge) | Ceased all use of portable gauges and placed them in secure storage until correction of all non-compliances to the satisfaction of the CNSC. | August 26 |
| June 20 (Laval, Québec) | Groupe ABS Inc. (Portable gauge) | Prohibited one worker from working with portable gauges until the worker was retrained and able to demonstrate safe work practices to CNSC’s satisfaction. | August 10 |
| August 18 (Edmonton, Alberta) | WSP Canada Inc. (Portable gauge) | Ceased using portable gauges at one base of operations and placed the radiation devices in secure storage until all items of non-compliance were corrected to the satisfaction of the CNSC. | September 28 |
| August 18 (Kelowna, British Columbia) | Arthon Industries Ltd. (Portable gauge) | Ceased use of all radiation devices and placed them in secure storage until all workers who were using the radiation devices have been trained in the transportation of Class 7 dangerous goods, until the licensee’s radiation protection program was implemented effectively, and until all non-compliances were corrected to the satisfaction of the CNSC. | November 8 |
| August 18 (Chelmsford, Ontario) | R.M. Belanger Ltd. (Portable gauge) | Removed two individuals from work involving portable gauges until they completed training on the licensee’s radiation safety program and demonstrated that they were working in accordance with CNSC regulations.Removed a portable gauge from service until necessary repairs were complete.Corrected all items of non-compliance to the satisfaction of the CNSC. | October 31 |
| August 24 (Lachine, Québec) | Isologic Innovative Radiopharmaceuticals Ltd. | Ceased servicing a cyclotron until an application for a new servicing licence was submitted, and a licence issued by the CNSC. | |
| September 15 (Kamloops, British Columbia) | Dawson Construction Ltd. (Portable gauge) | Ceased all use of radiation devices until the implementation of their radiation protection program and correction of non-compliances to the satisfaction of the CNSC. | September 30 |
| September 21 (Grand Cache, Alberta) | Milner Power Inc. (Fixed gauge) | Ceased all vessel entries into confined spaces with radiation devices and ceased performing any work that involved direct handling of the radiation devices until compliant procedures were submitted to and accepted by the CNSC, and the licensee’s workers were trained on these procedures. | December 9 |
| October 19 (Meadowbank, Nunavut) | Agnico-Eagle Mines Ltd. (Fixed gauge) | Ceased all vessel entries into confined spaces with radiation devices and ceased performing any work that involved direct handling of the radiation devices until compliant procedures were submitted to and accepted by the CNSC, and the licensee’s workers were trained on these procedures. | November 16 |
| October 26 (North Vancouver, British Columbia) | Horizon Engineering Inc. (Portable gauge) | Ceased using all portable gauges and placed these gauges in secure storage until the implementation of the licensee’s radiation protection program and correction of all items of non-compliance to the satisfaction of the CNSC. | November 24 |
| Issue date and location | Licensee or individual | Reason for issuing AMP | Penalty amount | Closure date |
|---|---|---|---|---|
| February 4 (Aurora, Ontario) | Nasirruddin Engineering Ltd. | Failure to keep radiation exposure to persons as low as reasonably achievable through the implementation of management control over work practices. | $3,730 | May 18 |
| February 24 (Ottawa, Ontario) | City of Ottawa | Failure to ensure that a radiation device was in the proper shielded condition when preparing it for transport and, once aware of that fact, failure to immediately notify the CNSC as required. | $7,930 | March 16 |
| May 2 (Merlin, Ontario) | Nuclear Services Canada | Transporting nuclear substances in a package that did not meet the applicable regulatory requirements. | $6,460 | June 9 |
| September 7 (Vernon, British Columbia) | Hoban Equipment Inc. | Carrying on a prescribed activity – i.e., the use of portable gauges – contrary to the licence issued to the company. | $10,360 | October 3 |
| September 9 (Grande Cache, Alberta) | Milner Power Inc. | Failure to comply with a licence condition for fixed gauge use in relation to entering confined spaces where radiation devices are present. | $3,970 | November 30 |
| September 29 (Onaping, Ontario) | Glencore Canada Corporation | Failure to follow the approved standard operating procedure for a maintenance activity. These actions led to a worker receiving a radiation dose. | $1,000 | October 4 |
| November 18 (Chelmsford, Ontario) | R.M. Belanger Ltd. | Failure to keep exposure to radiation to persons as low as reasonably achievable through implementation of management control over work practices. | $3,730 | November 30 |
| November 23 (Kelowna, British Columbia) | Interior Health Authority – Kelowna General Hospital | Failure to keep a record of all nuclear substances in their possession. | $1,000 | December 21 |
Appendix D: List of reported events in 2016
Table 8 includes all reported events by licensees in 2016, categorized using the International Nuclear and Radiological Event Scale (INES) tool.
| # | Date | INES rating | Type | Sector | Event summary |
|---|---|---|---|---|---|
| 2672 | January 3 | 0 | Packaging and transport | Medical | A package was delivered to the wrong licensee. |
| 2676 | January 7 | 0 | Malfunctioning device | Industrial | A certified exposure device operator was unable to retract the source to the fully shielded position and determined that the source assembly had become disconnected. He was able to return the source to the shielded position. |
| 2677 | January 8 | 0 | Missing or found | Academic | A licensee reported the loss of sealed sources when he was unable to account for their exact location. |
| 2678 | January 12 | 0 | Damaged device | Industrial | A certified exposure device operator tripped at a doorway while carrying an exposure device causing the device to drop to the ground. This damaged the device’s outer casing. The source remained locked in the exposure device and the radiation readings remained normal. |
| 2679 | January 6 | 0 | Damaged device | Industrial | An exposure device was being lowered by rope from a scaffold platform and fell approximately 1.5 m to the ground. There was damage to the front of device. The source remained locked in the exposure device and the radiation readings remained normal. |
| 2682 | January 15 | 0 | Packaging and transport | Industrial | A vehicle carrying an exposure device was involved in a collision. The device remained secure in the truck with no indication of damage. |
| 2684 | January 19 | 0 | Malfunctioning device | Academic | A worker was unable to remove a survey meter from the gamma checker gauge and the source control crank was difficult to turn. The radiation readings remained normal and the device was removed from service. |
| 2687 | January 14 | 0 | Malfunctioning device | Industrial | During a lockout procedure, a worker was unable to completely close the shutter of a radiation device. There was no visible physical damage and no abnormal readings were detected. The device was removed from service until it was repaired. |
| 2688 | January 19 | 0 | Malfunctioning device | Industrial | The shutter on a portable gauge was discovered in the open position after being transported for maintenance. There was no radiological exposure as a result of the event. |
| 2690 | January 22 | 0 | Packaging and transport | Industrial | During transport, a Type A package containing a cesium-137 source fell through the wooden floor of a trailer. The package sustained some damaged, but the source remained inside the package and was not damaged. |
| 2691 | January 20 | 0 | Malfunctioning device | Industrial | The shutter on a portable gauge was discovered in the open position after being transported and transferred for maintenance. There was no radiological exposure as a result of the event. |
| 2693 | January 25 | 0 | Packaging and transport | Academic | A package containing nuclear substances was not labelled as containing nuclear substances. |
| 2694 | January 27 | 0 | Spill | Commercial | A spill of a nuclear substance (I-131) occurred during the preparation of radioisotopes. The spill was covered with a steel plate. There was no skin contamination or thyroid intake as a result of this spill. |
| 2695 | January 29 | 0 | Spill | Commercial | A vial containing nuclear substances (I-131) cracked upon removal from the package and leaked into the lead pot. |
| 2696 | February 2 | 0 | Packaging and transport | Medical | A package containing 370 megabecquerels (MBq) of iodine-131 was delivered to the wrong hospital. The hospital arranged for a courier to deliver the package to the correct location. |
| 2699 | January 16 | 0 | Breach of security | Medical | A security guard found a room unlocked and the intrusion alarm unarmed while performing a security sweep. A new housekeeper was not aware of the requirements to arm the system and lock the door after cleaning. |
| 2704 | February 8 | 0 | Spill | Medical | A vial was dropped during the preparation of a radiopharmaceutical resulting in a spill (I-131). The spill was contained on a rubber stress mat and was bagged and held for decay. There was no skin contamination as a result of this spill. |
| 2707 | February 7 | 0 | Breach of security | Medical | Keys for rooms containing prescribed equipment were reported stolen. |
| 2712 | February 22 | 0 | Packaging and transport | Commercial | A Type A package containing a vial of iodine-131 was delivered to a different hospital than intended. The licensee that received the package held a valid CNSC licence. The package was delivered to correct licensee the next day. |
| 2714 | February 27 | 0 | Damaged device | Industrial | The remote control of an exposure device was damaged after coming in contact with very hot surface. |
| 2716 | February 29 | 0 | Malfunctioning device | Industrial | A fixed gauge was not indicating that the shutter was closed when the shutter should have been closed. It was determined that the shutter was closed, but the computer communication was malfunctioning. |
| 2718 | March 4 | 0 | Damaged device | Industrial | A portable gauge was damaged by a compactor at a construction site. |
| 2728 | March 18 | 0 | Malfunctioning device | Industrial | The shutter on a portable gauge was discovered in the open position. The gauge was stored 15 m away from any occupied area. There was no radiological exposure as a result of the event. |
| 2729 | February 4 | 0 | Breach of security | Academic | An unauthorized person was working in dedicated radiation zone. |
| 2730 | March 3 | 0 | Unplanned exposure | Industrial | An unplanned exposure of a member of the public occurred when they entered an area where radiography work was taking place. |
| 2731 | March 24 | 0 | Malfunctioning device | Industrial | The shutter on a portable gauge was discovered in the open position after being transported and transferred following maintenance. |
| 2732 | March 22 | 0 | Damaged device | Indutrial | An exposure device was being transported on top of a wheeled toolbox and fell approximately 1 m to the ground. There was no visible damage to the device. The device was sent for maintenance and was confirmed to be undamaged. |
| 2735 | March 30 | 0 | Packaging and transport | Medical | A package containing a sealed source was reported as having a higher dose rate than expected for the source contained inside, although the dose rate measured on the package remained within the regulatory limits. It was determined that the source was not fully inserted into the shielded position. |
| 2736 | February 29 | 0 | Damaged device | Industrial | An exposure device fell to the ground while being lowered from scaffolding. Some external damaged to the device was reported. Leak test results were negative. |
| 2740 | April 4 | 0 | Damaged device | Industrial | An exposure device fell approximately 1.3 m to the ground from scaffolding. Leak tests were negative. |
| 2742 | April 29 | 0 | Packaging and transport | Medical | A package containing therapeutic iodine-131 was delivered to the wrong licensee. The package was picked up and delivered by the carrier to the correct recipient on the same day. |
| 2746 | January 13 | 0 | Malfunctioning device | Industial | A shutter on a fixed gauge was reported as being stuck due to corrosion. The service provider repaired the gauge in place and a protection over the gauge was installed to prevent recurrence. |
| 2747 | March 18 | 0 | Spill | Medical | A spill of a nuclear substance (Tc-99m) occurred during the manipulation of a vial of a medical isotope. There was no skin contamination as a result. |
| 2748 | April 4 | 0 | Packaging and transport | Industrial | A Type A package was damaged during transport. The device inside was not damaged. |
| 2749 | April 19 | 0 | Unplanned exposure | Indutrial | An unplanned exposure to a member of the public occurred when they entered an area where radiography work was taking place. |
| 2751 | April 21 | 0 | Spill | Medical | A spill of a nuclear substance (Tc-99m) occurred in a microwave in a designated laboratory. There was no skin contamination as a result of this event. |
| 2757 | May 8 | 0 | Malfunctioning device | Industrial | Shutters on three fixed gauges were stuck in the open position. The devices were kept in the normal operating locations to prevent exposure to persons. The three devices underwent maintenance to fix the shutters. |
| 2758 | May 9 | 0 | Packaging and transport | Commercial | A package was delivered to the wrong licensee. The package contained a rubidium/strontium-82 generator. The package was picked up and delivered by the carrier to the correct recipient on the same day. |
| 2759 | May 12 | 0 | Malfunctioning device | Industrial | The shutter and shutter handle on a fixed gauge was not operating properly. The handle was repaired. |
| 2760 | May 23 | 0 | Missing or found | Indutrial | A vehicle containing a portable gauge was reported stolen. The vehicle and portable gauge were recovered. |
| 2762 | May 30 | 0 | Damaged device | Industrial | The guide tube of an exposure device was damaged when a pipe fell on it. The source was successfully retracted into the shielded position within the exposure device |
| 2764 | June 7 | 0 | Unplanned exposure | Indutrial | An unplanned exposure to a member of the public occurred when they entered an area where radiography work was taking place. |
| 2766 | June 17 | 0 | Damaged device | Industrial | A portable gauge was damaged when hit by the drill rod and scraper plate. A leak test was performed and the results were negative. |
| 2769 | June 15 | 0 | Missing or found | Medical | A therapeutic dose of iodine-131 was reported as missing following delivery. |
| 2770 | June 17 | 0 | Damaged device | Academic | A radiation device was hit by a crane. A leak test was performed and the results were negative. |
| 2771 | June 21 | 0 | Spill | Commercial | A spill of a nuclear substance (In-111) occurred when a worker was disposing of analytical samples in a designated area. There were no measurable dose rates as a result of the incident. |
| 2775 | June 28 | 0 | Missing or found | Medical | A dew pointer was found in a load of scrap metal. |
| 2776 | June 1 | 0 | Packaging and transport | Industrial | A radiation device was reported as lost during transport. The device was located by the carrier a week later. |
| 2777 | June 25 | 0 | Packaging and transport | Commercial | The external cardboard package of a shipment was damaged by water. There was no damage to the internal packaging and no leaks or external contamination were detected. |
| 2779 | June 23 | 0 | Packaging and transport | Commercial | A package was forgotten in the parking lot of a hospital by a driver. The licensee sent someone to retrieve the package from the parking lot. |
| 2780 | July 6 | 0 | Damaged device | Industrial | A portable gauge was damaged when run over by a vehicle at a construction site. The handle broke, but the source was able to be retracted to the shielded position |
| 2781 | June 27 | 0 | Packaging and transport | Academic | A package was reported as contaminated above the regulatory limit for external contamination |
| 2782 | July 14 | 0 | Spill | Commercial | A spill of a nuclear substance (Tc-99m) occurred during the preparation of radioisotopes. The floor where the spill occurred was decontaminated. |
| 2784 | July 12 | 0 | Damaged device | Industrial | A portable gauge was damaged when hit by an excavator. The handle was bent. A leak test was performed and the results were negative. |
| 2786 | July 19 | 0 | Unplanned exposure | Industrial | A worker entered a vessel to perform maintenance work while the fixed gauges mounted on the vessel were still in the open position. The worker received a dose below the effective dose limit for members of the public. |
| 2787 | July 21 | 0 | Damaged device | Industrial | A portable gauge was damaged when it fell off the back of a truck. The sources remained secured within device. A leak test was performed and the results were negative. |
| 2791 | July 24 | 0 | Packaging and transport | Industrial | A vehicle transporting a portable gauge was involved in a collision. There was no visible damage to the package or gauge, and the radiation readings remained normal. |
| 2792 | July 25 | 0 | Packaging and transport | Industrial | A vehicle transporting a portable gauge was involved in a collision. There was no visible damage to the package or gauge, and the radiation readings remained normal. |
| 2793 | August 3 | 0 | Missing or found | Industrial | A licensee reported the loss of an exposure device while transporting it to a job site. The exposure device was recovered the following day. |
| 2795 | July 15 | 0 | Packaging and transport | Industrial | A portable gauge was misplaced during transport. The carrier later found the package and delivered it to the consignee. |
| 2796 | July 29 | 0 | Packaging and transport | Commercial | Two Type A packages were misplaced during transport. The carrier found the packages in a warehouse. |
| 2797 | July 19 | 0 | Damaged device | Industrial | A portable gauge was damaged when hit by an excavator on a construction site. The source rod was damaged but could be retracted into the shielded position. The licensee made arrangements for the disposal of the device. |
| 2802 | July 8 | 0 | Packaging and transport | Industrial | A vehicle carrying an exposure device was involved in a collision. There was no damage to the exposure device. A leak test was performed and the results were negative. |
| 2807 | August 28 | 0 | Unplanned exposure | Commercial | A nuclear energy worker (NEW) was exposed to iodine-131 as a result of a needle stick. |
| 2810 | August 17 | 0 | Packaging and transport | Academic | A package sustained minor damage during transport. The external cardboard packaging was damaged. The inner components were not damaged. The licensee repacked them in another box. |
| 2811 | August 19 | 0 | Damaged device | Industrial | A fixed gauge was reported as damaged. The licensee confirmed that radiation readings remained normal. |
| 2812 | August 22 | 0 | Spill | Commercial | A spill of a nuclear substance (Tc-99m) occurred during the preparation of radiopharmaceuticals. |
| 2813 | August 15 | 0 | Malfunctioning device | Industrial | The shutter on a portable gauge was stuck in the open position. The shutter operated normally after being cleaned. |
| 2816 | August 25 | 0 | Packaging and transport | Commercial | A package was delivered to the incorrect location. Arrangements were made with the carrier to deliver it to the correct location. |
| 2823 | September 6 | 0 | Missing or found | Industrial | A portable gauge was stolen from a construction site. The police were notified and the portable gauge was recovered. |
| 2824 | September 8 | 0 | Packaging and transport | Industrial | A package was damaged during transport. There was no contamination or leakage as a result of this event. |
| 2826 | September 9 | 0 | Spill | Medical | A spill of a nuclear substance (F-18) occurred when a technician was administering a nuclear medicine dose to a patient. The technician’s skin was contaminated. The dose received was below the regulatory limit. |
| 2827 | August 30 | 0 | Damaged device | Industrial | A portable gauge was damaged at a construction site. A leak test was performed and the results were negative. |
| 2830 | September 8 | 0 | Packaging and transport | Commercial | A package was damaged as a result of heavy rain. There was no contamination or leakage as a result of this event. |
| 2834 | September 10 | 0 | Malfunctioning device | Industrial | The source in an exposure device failed to return to the shielded position. A source recovery operation was performed and the source was returned to the fully shielded position. |
| 2836 | August 24 | 0 | Packaging and transport | Industrial | A vehicle transporting a portable gauge was involved in a collision. There was no damage to the device or its package. |
| 2838 | September 9 | 0 | Malfunctioning device | Industrial | The shutter on a fixed gauge was not closing. Since the device could not be repaired, the licensee made arrangements for its disposal. |
| 2839 | September 16 | 0 | Spill | Medical | A spill of a nuclear substance (Tc-99m) occurred while administering a nuclear medicine dose. The NEW received a dose to the skin below regulatory limits as a result of the incident. |
| 2840 | September 9 | 0 | Spill | Medical | A spill of a nuclear substance (F-18) occurred when a technician was administering a nuclear medicine dose. The technician received a dose to the skin that was below the regulatory limits. |
| 2841 | September 14 | 0 | Spill | Commercial | A spill of a nuclear substance (I-131) occurred during the preparation of radiopharmaceuticals. There was no skin contamination or thyroid uptake as a result of the incident. |
| 2842 | September 15 | 0 | Damaged device | Industrial | A worker noticed that a portable gauge had minor damage. The device was removed from use and a leak test performed. The results were negative. |
| 2843 | August 22 | 0 | Unplanned exposure | Medical | Workers accessed the roof of a linear accelerator bunker while the device was in operation. |
| 2844 | September 20 | 0 | Packaging and transport | Commercial | A package sustained minor damaged during transport. The material was repacked and shipped back to the consignor. |
| 2845 | September 23 | 0 | Damaged device | Industrial | A portable gauge was damaged at a construction site. There was no contamination or leakage as a result of this event. |
| 2846 | September 20 | 0 | Packaging and transport | Industrial | A vehicle transporting a portable gauge was involved in a collision. The package and gauge were undamaged. There was no contamination or leakage as a result of this event. |
| 2848 | May 15 | 0 | Damaged device | Industrial | A portable gauge was damaged at a construction site. There was no contamination or leakage as a result of this event. |
| 2850 | September 22 | 0 | Damaged device | Industrial | A portable gauge was damaged at a construction site. There was no contamination or leakage as a result of this event. |
| 2852 | September 19 | 0 | Worker injured | Industrial | A portable gauge worker was at a job site preparing to use the portable gauge when his truck was hit by a construction vehicle. The worker was injured and taken to hospital. The device fell to the ground, but was not damaged. |
| 2854 | September 15 | 0 | Damaged device | Industrial | An exposure device was dropped 18 m. The device was taken out of service and was inspected by a servicing company. |
| 2856 | September 27 | 0 | Damaged device | Industrial | A portable gauge was damaged at a construction site. There was no contamination or leakage as a result of this event. |
| 2857 | September 27 | 0 | Spill | Medical | A spill of a nuclear substance (Y-90) occurred during the administration of a nuclear medicine therapy. The spill was contained and the contaminated working materials and personal protective equipment were isolated. |
| 2859 | September 29 | 0 | Breach of security | Industrial | A break-in occurred at a portable gauge licensee. However, the storage bunker where the gauges were located was not tampered with. No devices were stolen. |
| 2860 | September 22 | 0 | Unplanned exposure | Industrial | An unplanned exposure to a member of the public occurred when they entered an area where radiography work was taking place. |
| 2861 | September 23 | 0 | Packaging and transport | Industrial | A vehicle transporting a portable gauge was involved in a collision. The device was not damaged. |
| 2862 | September 29 | 0 | Packaging and transport | Commercial | A vial of fluorine-18 was broken upon receipt of a package. The contents were contained in a lead pot. There was no external contamination or leakage as a result of this event. |
| 2864 | September 30 | 0 | Spill | Medical | A nuclear substance (F-18) spilled into its lead pot during transport through the nuclear medicine department. There was no contamination or leakage outside the lead pot. |
| 2866 | October 4 | 0 | Missing or found | Industrial | A car transporting a portable gauge was reported as missing. It was determined that the car had been towed to a city impound lot. The device and car were recovered. |
| 2867 | October 4 | 1 | Missing or found | Industrial | A portable gauge was stolen from a car parked at a private residence. The device has not been recovered. |
| 2868 | October 4 | 0 | Packaging and transport | Medical | A vehicle carrying medical isotopes was involved in a collision. There was no damage to the package. There was no contamination or leakage as a result of this event. |
| 2869 | October 4 | 0 | Missing or found | Commercial | A piece of metal with a radiation warning sign was discovered at a scrap metal facility. |
| 2878 | September 26 | 0 | Unplanned exposure | Commercial | A NEW received a needle stick and was potentially exposed to fluorine-18. The resulting dose was below the regulatory limits. |
| 2879 | October 13 | 0 | Malfunctioning device | Industrial | The shutter on a fixed gauge would not close. A contractor was brought in to remove the gauge from its location and to service it. |
| 2880 | October 18 | 0 | Spill | Medical | A spill of a nuclear substance (Tc-99m) occurred during the preparation of a nuclear medicine dose. The technician’s skin was contaminated and the dose received was below the regulatory limits. |
| 2881 | October 20 | 0 | Unplanned exposure | Commercial | A NEW received skin contamination by technetium-99m. The estimated dose was determined to be below the regulatory limits. |
| 2882 | October 10 | 0 | Malfunctioning device | Industrial | The source of a fixed gauge could not be retracted into the shielded position. The radiation readings remained normal. |
| 2883 | October 21 | 0 | Missing or found | Academic | A nickel-63 sealed source was discovered missing from a gas chromatograph. The source was recovered. |
| 2885 | October 21 | 0 | Packaging and transport | Industrial | A portable gauge, in its Type A package, fell off the back of a vehicle at a construction site. The package was damaged, but the device was not. The radiation readings remained normal as a result of this incident. |
| 2886 | October 24 | 0 | Spill | Commercial | A spill of a nuclear substance (Sr-82) occurred when a waste container overflowed. The contaminated laboratory equipment was removed and placed in storage for decay. There was no personnel contamination as a result of the incident. |
| 2892 | October 24 | 0 | Packaging and transport | Academic | A package containing 163 MBq of lutitum-177 was shipped as an excepted package, empty package instead of as a Type A package. The package used was a Type A package. |
| 2896 | October 27 | 0 | Packaging and transport | Industrial | A vehicle transporting an exposure device was involved in a collision. There was no damage to the device as a result of this incident. |
| 2898 | November 3 | 0 | Packaging and transport | Commercial | A package containing medical isotopes was lost during transport. It was recovered later in the day and delivered the following day. |
| 2627 | November 10 | 0 | Missing or found | Industrial | A portable gauge that was stolen on November 7, 2015 was recovered |
| 2899 | October 26 | 0 | Packaging and transport | Academic | A package containing phosphorus-32 had substantial damage to the bottom of the package. The lead pot inside the package was intact. There was no contamination or leakage as a result of this event. |
| 2901 | November 1 | 0 | Malfunctioning device | Industrial | A portable gauge with an open shutter was found in a storage area. The licensee was able to close the shutter. |
| 2906 | November 8 | 0 | Unplanned exposure | Industrial | Two contractors entered a vessel while the fixed gauges mounted on the vessel were still in the open position. The estimated dose received was below regulatory limits. |
| 2907 | November 16 | 0 | Spill | Commercial | A spill of a nuclear substance (Tc-99m) occurred when a vial broke during the manufacturing of medical isotopes. The area where the spill occurred was identified and access was restricted. |
| 2909 | November 16 | 0 | Damaged device | Industrial | A portable gauge was damaged at construction site. There was no contamination or leakage and the radiation readings remained normal as a result of this event. |
| 2911 | November 23 | 0 | Packaging and transport | Industrial | A vehicle transporting a portable gauge was involved in a collision. There was no damage to the device or its package. |
| 2913 | November 25 | 0 | Packaging and transport | Medical | Packages containing medical isotopes were found with external contamination above the regulatory limit. |
| 2915 | November 28 | 0 | Packaging and transport | Industrial | A vehicle transporting a portable gauge was involved in a collision. There was no damage to the device or its package. |
| 2918 | November 30 | 0 | Spill | Commercial | A spill of a nuclear substance (I-131) occurred during the manufacture of medical isotopes and limited to the vented, shielded glove box. |
| 2919 | December 1 | 0 | Packaging and transport | Industrial | A vehicle transporting a portable gauge was involved in a collision. There was no damage to the device or its package. |
| 2920 | October 28 | 0 | Spill | Medical | A spill of a nuclear substance (Y-90) occurred during the administration of a nuclear medicine therapy. The incident resulted in skin contamination of the technician with an estimated dose above the regulatory limit. |
| 2921 | December 2 | 1 | Missing or found | Industrial | A portable gauge was stolen from a truck parked outside a private residence. The device has not been recovered. |
| 2922 | November 16 | 0 | Unplanned exposure | Industrial | An unplanned exposure occurred when a welder was present in an area where industrial radiography was being conducted. The individual received a dose that was below regulatory limits. |
| 2925 | December 21 | 0 | Missing or found | Medical | A Category 5 source (i.e., a cancer seed) was reported as lost. |
| 2926 | December 8 | 0 | Packaging and transport | Commercial | Two packages were reported as lost during transport. The packages were located by the carrier four days later. |
| 2928 | December 7 | 0 | Damaged device | Industrial | A damaged exposure device was found in a storage area. Radiation readings remained normal. The device was removed from service and sent for maintenance. |
| 2929 | December 1 | 0 | Breach of security | Medical | An access card for a high-level laboratory area was found in a public washroom. The security staff for the facility disabled the card. |
| 2932 | December 9 | 0 | Damaged source | Medical | A radiotherapy seed was cut in half during preparation of tissue sample. No contamination resulted from the incident. |
| 2933 | December 10 | 0 | Unplanned exposure | Industrial | An unplanned exposure occurred when a worker performing maintenance work entered a vessel mounted with fixed gauges while they were in the open position. The dose received was below regulatory limits. |
| 2937 | December 4 | 0 | Malfunctioning device | Industrial | The shutter on a fixed gauge was not working properly. The gauge was sent for repair. |
| 2938 | December 20 | 0 | Malfunctioning device | Industrial | The shutter on a fixed gauge was not working properly. The shutter was cleaned and repaired. |
| 2941 | December 15 | 0 | Packaging and transport | Industrial | A vehicle transporting a portable gauge was involved in a collision. There was no damage to the device or its package. |
| 2942 | December 21 | 0 | Spill | Commercial | A spill of a nuclear substance (F-18) occurred within a lab resulting in skin contamination of a worker. The laboratory was closed and isolated, and the worker was decontaminated. |
| 2943 | October 23 | 0 | Packaging and transport | Industrial | A vehicle transporting a portable gauge was involved in a collision. There was no damage to the device or its package. |
| 2944 | December 22 | 0 | Packaging and transport | Commercial | A vehicle transporting medical isotopes was involved in a collision. The packages being transported were not damaged as a result of the incident. |
| 2981 | December 21 | 0 | Packaging and transport | Industrial | A vehicle transporting an exposure device was involved in a collision. The device was not damaged as a result of the incident. |
| 3050 | February 21 | 0 | Packaging and transport | Commercial | A Type A package containing medical isotopes was damaged during transport. Since the damage was minor, and there was no leakage, the package was allowed to continue in transport. |
Appendix E: Inspections conducted in 2016
Note: Many inspections for Class II facilities are of consolidated licences. These are shown only once in the table.
| Inspection date | Licensee name | City | Province | Inpection type | Sector |
|---|---|---|---|---|---|
| 4-Jan-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| 5-Jan-16 | Echo NDE Inc. | Red Deer | AB | Type II | Industrial |
| 5-Jan-16 | Mistras Services Inc. | Lévis | QC | Type II | Industrial |
| 5-Jan-16 | The Scarborough Hospital | Scarborough | ON | Type II | Medical |
| 5-Jan-16 | The Scarborough Hospital | Scarborough | ON | Type II | Medical |
| 5-Jan-16 | The Scarborough Hospital | Scarborough | ON | Type II | Medical |
| 5-Jan-16 | The Scarborough Hospital | Scarborough | ON | Type II | Medical |
| 6-Jan-16 | 2345171 Ontario Inc. | Guelph | ON | Type II | Medical |
| 6-Jan-16 | Brant Community Healthcare System | Brantford | ON | Type II | Medical |
| 6-Jan-16 | Brant Community Healthcare System | Brantford | ON | Type II | Medical |
| 6-Jan-16 | Mistras Services | Lévis | QC | Type II | Industrial |
| 7-Jan-16 | CMT Engineering Inc. | St. Clemens | ON | Type II | Industrial |
| 7-Jan-16 | The Chemours Canada Company | Maitland | ON | Type II | Industrial |
| 7-Jan-16 | Welltec Canada Inc. | Stettler | AB | Type II | Industrial |
| 8-Jan-16 | ARCADIS Canada Inc. | Richmond Hill | ON | Type II | Industrial |
| 8-Jan-16 | ARCADIS Canada Inc. | Richmond Hill | ON | Type II | Industrial |
| 8-Jan-16 | Cornwall Community Hospital | Cornwall | ON | Type II | Medical |
| 8-Jan-16 | Cornwall Community Hospital | Cornwall | ON | Type II | Medical |
| 8-Jan-16 | Lantheus MI Canada Inc. | Montéal | QC | Type II | Commercial |
| 11-Jan-16 | Centre for Probe Development and Commercialization | Hamilton | ON | Type II | Commercial |
| 11-Jan-16 | Geolog Solutions Inc. | Red Deer County | AB | Type II | Industrial |
| 11-Jan-16 | Inter Medico, Division of | Markham | ON | Type II | Commercial |
| 13-Jan-16 | Brampton Nuclear Services | Brampton | ON | Type II | Medical |
| 13-Jan-16 | Canadian Tire Corporation, Ltd. | Toronto | ON | Type II | Commercial |
| 13-Jan-16 | Centre de santé et de services sociaux de la Haute-Yamaska | Granby | QC | Type II | Medical |
| 13-Jan-16 | Centre de santé et de services sociaux de la Haute-Yamaska | Granby | QC | Type II | Medical |
| 13-Jan-16 | Ezeflow Inc. | Granby | QC | Type II | Industrial |
| 13-Jan-16 | Ezeflow Inc. | Granby | QC | Type II | Industrial |
| 13-Jan-16 | Isologic Innovative Radiopharmaceuticals Ltd. | Lachine | QC | Type II | Commercial |
| 13-Jan-16 | Maxxam Analytics International Corporation | Mississauga | ON | Type II | Academic & research |
| 13-Jan-16 | Uni-Tech Inspection Services Ltd. | South Glengarry | ON | Type II | Industrial |
| 13-Jan-16 | Vale Canada Ltd. | Copper Cliff | ON | Type II | Industrial |
| 14-Jan-16 | Bayer Inc. | Mississauga | ON | Type II | Commercial |
| 14-Jan-16 | Buffalo Inspection Services (2005) Inc. | Edmonton | AB | Type II | Industrial |
| 14-Jan-16 | Mistras Optimus Inc. | Saint-Lambert | QC | Type II | Industrial |
| 14-Jan-16 | Sartell Instrumentation Ltd. | Mississauga | ON | Type II | Commercial |
| 14-Jan-16 | Stuart Hunt & Associates Ltd. | St. Albert | AB | Type II | Commercial |
| 15-Jan-16 | Centre de santé et de services | Longeuil | QC | Type II | Medical |
| 15-Jan-16 | Centre de santé et de services sociaux Pierre-Boucher | Longeuil | QC | Type II | Medical |
| 15-Jan-16 | Mistras Services Inc. | Lévis | QC | Type II | Industrial |
| 18-Jan-16 | Bay Cardiac Diagnostic Inc. | Toronto | ON | Type II | Medical |
| 18-Jan-16 | Burton Enterprises Inc. | Fort Williams | NS | Type II | Industrial |
| 18-Jan-16 | Centre de santé et de services sociaux du Coeur-de-l'Ile | Montéal | QC | Type II | Medical |
| 18-Jan-16 | Centre de santé et de services sociaux du Coeur-de-l'Ile | Montréal | QC | Type II | Medical |
| 18-Jan-16 | Dr. Melanie Dara Hobbs, P. Eng. | Coldbrook | NS | Type II | Industrial |
| 18-Jan-16 | Tier 1 Energy Solutions Inc. | Calgary | AB | Type II | Industrial |
| 19-Jan-16 | Atomic NDT Ltd. | Edmonton | AB | Type II | Industrial |
| 19-Jan-16 | Buffalo Inspection Services (2005) | Edmonton | AB | Type II | Industrial |
| 19-Jan-16 | Candec Consultants Ltd. | Richmond Hill | ON | Type II | Industrial |
| 19-Jan-16 | Custom Fabricators & Machinists Ltd. / Fabricants et Mach | Saint John | NB | Type II | Industrial |
| 19-Jan-16 | Graham Bros. Construction Limited | Brampton | ON | Type II | Industrial |
| 19-Jan-16 | Graham Bros. Construction Limited | Brampton | ON | Type II | Industrial |
| 19-Jan-16 | Groupe Qualitas Inc. | Montréal | QC | Type II | Industrial |
| 19-Jan-16 | Groupe Vétéri Médic Inc. | Brossard | QC | Type II | Medical |
| 19-Jan-16 | Keyera Corp. | Calgary | AB | Type II | Industrial |
| 19-Jan-16 | Peterborough Regional Health Centre | Peterborough | ON | Type II | Medical |
| 19-Jan-16 | Peterborough Regional Health Centre | Peterborough | ON | Type II | Medical |
| 19-Jan-16 | Shaw Pipeline Services Ltd. | Sherwood Park | AB | Type II | Industrial |
| 19-Jan-16 | The Minute Maid Company Canada / La compagnie minute maid | Peterborough | ON | Type II | Industrial |
| 19-Jan-16 | TISI Canada Inc. | Oakville | ON | Type II | Industrial |
| 20-Jan-16 | Agrium | Fort Saskatchewan | AB | Type II | Industrial |
| 20-Jan-16 | Coca-Cola Refresments Canada Company | Brampton | ON | Type II | Industrial |
| 20-Jan-16 | Englobe Corp. | Laval | QC | Type II | Industrial |
| 20-Jan-16 | Englobe Corp. | Laval | QC | Type II | Industrial |
| 20-Jan-16 | Kawartha Diagnostic Imaging Ltd. | Peterborough | ON | Type II | Medical |
| 20-Jan-16 | Kawartha Diagnostic Imaging Ltd. | Peterborough | ON | Type II | Medical |
| 20-Jan-16 | Nortech Advanced N.D.T. Ltd. | Sherwood Park | AB | Type II | Industrial |
| 20-Jan-16 | Nova Scotia Health Authority | Halifax | NS | Type II | Commercial |
| 20-Jan-16 | Terraspec Engineering Inc. | Peterborough | ON | Type II | Industrial |
| 20-Jan-16 | TJ Inspection Services | Dartmouth | NS | Type II | Industrial |
| 21-Jan-16 | Acuren Group Inc. | Edmonton | AB | Type II | Industrial |
| 21-Jan-16 | Cambium Inc. | Peterborough | ON | Type II | Industrial |
| 21-Jan-16 | East Coast Veterinary Group Limited | Peterborough | ON | Type II | Medical |
| 21-Jan-16 | Engtec Consulting Inc. | Vaughan | ON | Type II | Industrial |
| 21-Jan-16 | Gamma-Tech Inspection Ltd. | Calgary | AB | Type II | Industrial |
| 21-Jan-16 | Gamma-Tech Inspection Ltd. | Calgary | AB | Type II | Industrial |
| 21-Jan-16 | Gamma-Tech Inspection Ltd. | Calgary | AB | Type II | Industrial |
| 21-Jan-16 | Hôpital Santa Cabrini | Montréal | QC | Type II | Medical |
| 21-Jan-16 | Hôpital Santa Cabrini | Montréal | QC | Type II | Medical |
| 21-Jan-16 | Hunt Inspection Ltd. | Stettler | AB | Type II | Industrial |
| 21-Jan-16 | IRISNDT Corp. | Edmonton | AB | Type II | Industrial |
| 21-Jan-16 | J & P Levesque Bros Haulage Ltd. | Bancroft | ON | Type II | Industrial |
| 21-Jan-16 | Kingston General Hospital | Kingston | ON | Type I | Medical |
| 21-Jan-16 | LEA Consulting Ltd. | Markham | ON | Type II | Industrial |
| 21-Jan-16 | Les Inspections Thermetco Inc. | Montréal | QC | Type II | Industrial |
| 21-Jan-16 | Northumberland Hills Hospital | Coburg | ON | Type II | Medical |
| 21-Jan-16 | Stuart Hunt & Associates Ltd. | St. Albert | AB | Type II | Commercial |
| 21-Jan-16 | TJ Inspection Services | Dartmouth | NS | Type II | Industrial |
| 22-Jan-16 | Conquest Engineering Ltd. | Saint John | NB | Type II | Industrial |
| 22-Jan-16 | Izaak Walton Killam Health Centre | Halifax | NS | Type II | Medical |
| 22-Jan-16 | Scanning Technolgies Inc. | Sherwood Park | AB | Type II | Industrial |
| 22-Jan-16 | Spectrum NDT Ltd. | Calgary | AB | Type II | Industrial |
| 25-Jan-16 | Centre hospitalier universitaire de Sherbrooke | Sherbrooke | QC | Type I | Medical |
| 25-Jan-16 | GHD Consultants Ltd. | Saint-Laurent | QC | Type II | Industrial |
| 25-Jan-16 | GHD Consultants Ltd. | Saint-Laurent | QC | Type II | Industrial |
| 26-Jan-16 | Capital Paving Inc. | Guelph | ON | Type II | Industrial |
| 26-Jan-16 | K.V. Inspection Services Ltd. | Oakville | ON | Type II | Industrial |
| 27-Jan-16 | Centre de santé et des services sociaux Pierre-de-Saurel | Sorel | QC | Type II | Medical |
| 27-Jan-16 | Centre de santé et des services sociaux Pierre-de-Saurel | Sorel-Tracy | QC | Type II | Medical |
| 27-Jan-16 | Englobe Corp. | Laval | QC | Type II | Industrial |
| 27-Jan-16 | Honeywell-Measurex Inc. | Lachine | QC | Type II | Commercial |
| 27-Jan-16 | K.V. Inspection Services Ltd. | Oakville | ON | Type II | Industrial |
| 27-Jan-16 | Sunnybrook Health Sciences Centre | Toronto | ON | Type II | Academic & research |
| 27-Jan-16 | Sunnybrook Health Sciences Centre | Toronto | ON | Type II | Academic & research |
| 27-Jan-16 | Sunnybrook Health Sciences Centre | Toronto | ON | Type II | Medical |
| 27-Jan-16 | Sunnybrook Health Sciences Centre | Toronto | ON | Type II | Medical |
| 27-Jan-16 | TISI Canada Inc. | Oakville | ON | Type II | Industrial |
| 28-Jan-16 | Labatt Bewing Comapny Ltd. / La Brasserie Labatt limitée | London | ON | Type II | Industrial |
| 28-Jan-16 | Schlumberger Canada Ltd. | Calgary | AB | Type II | Industrial |
| 28-Jan-16 | Siemens Canada Ltd. | Oakville | ON | Type II | Commercial |
| 28-Jan-16 | Sunnybrook Health Sciences Centre | Toronto | ON | Type II | Medical |
| 28-Jan-16 | Sunnybrook Health Sciences Centre | Toronto | ON | Type II | Medical |
| 28-Jan-16 | Sunnybrook Health Sciences Centre | Toronto | ON | Type II | Medical |
| 29-Jan-16 | Kubota Materials Canada Corp. | Orillia | ON | Type II | Industrial |
| 29-Jan-16 | Kubota Materials Canada Corp. | Orillia | ON | Type II | Industrial |
| 1-Feb-16 | Atomic NDT Ltd. | Edmonton | AB | Type II | Industrial |
| 1-Feb-16 | Valmet Ltée / Valmet Ltd. | Ste-Anne-des-Plaines | QC | Type II | Industrial |
| 1-Feb-16 | West End 2425 Nuclear Medicine Inc. | Toronto | ON | Type II | Medical |
| 2-Feb-16 | C.B Non-destructive Testing Ltd. | Oakville | ON | Type II | Industrial |
| 2-Feb-16 | Canberra Co. | Concord | ON | Type II | Commercial |
| 2-Feb-16 | Canberra Co. | Concord | ON | Type II | Commercial |
| 2-Feb-16 | DGI Geoscience Inc. | Barrie | ON | Type II | Industrial |
| 2-Feb-16 | Groupe Vétéri Médic Inc. | Brossard | QC | Type II | Medical |
| 2-Feb-16 | Innotech Inspection Soultions Ltd. | Sherwood Park | AB | Type II | Industrial |
| 2-Feb-16 | Insight Inspections Inc. | Edmonton | AB | Type II | Industrial |
| 2-Feb-16 | Perfection Inspection Ltd. | Cambridge | ON | Type II | Industrial |
| 2-Feb-16 | Thermo Gamma-Metrics LLC | San Diego | CA | Type II | Commercial |
| 2-Feb-16 | Trillium Health Partners | Mississauga | ON | Type II | Medical |
| 2-Feb-16 | Trillium Health Partners | Mississauga | ON | Type II | Medical |
| 3-Feb-16 | CIUSSS de l'Est-de-l'Île-de-Montréal | Montréal | QC | Type II | Medical |
| 3-Feb-16 | IRISNDT Corp. | Edmonton | AB | Type II | Industrial |
| 4-Feb-16 | Contro Valve Equipment Inc. | Burlington | ON | Type II | Commercial |
| 4-Feb-16 | Hôpital Montfort | Ottawa | ON | Type II | Medical |
| 4-Feb-16 | Insight Medical Holdings Ltd. | Edmonton | AB | Type II | Medical |
| 4-Feb-16 | Northern Alberta Institute of Technology | Edmonton | AB | Type II | Industrial |
| 4-Feb-16 | Seymour Pacific Developments Ltd. | Regina | SK | Type II | Industrial |
| 4-Feb-16 | South Rock Ltd. | Calgary | AB | Type II | Industrial |
| 4-Feb-16 | The Graff Company Ltd. | Mississauga | ON | Type II | Industrial |
| 4-Feb-16 | TISI Canada Inc. | Oakville | ON | Type II | Industrial |
| 4-Feb-16 | Université du Québec à Montréal | Montréal | QC | Type II | Academic & research |
| 4-Feb-16 | William Osler Health Centre | Brampton | ON | Type II | Medical |
| 4-Feb-16 | William Osler Health Centre | Brampton | ON | Type II | Medical |
| 5-Feb-16 | Step Energy service Ltd. | Red Deer | AB | Type II | Industrial |
| 5-Feb-16 | The Ottawa Hospital | Ottawa | ON | Type II | Commercial |
| 5-Feb-16 | The Ottawa Hospital | Ottawa | ON | Type II | Commercial |
| 5-Feb-16 | The Ottawa Hospital | Ottawa | ON | Type II | Commercial |
| 5-Feb-16 | The Ottawa Hospital | Ottawa | ON | Type II | Medical |
| 5-Feb-16 | The Ottawa Hospital | Ottawa | ON | Type II | Medical |
| 5-Feb-16 | The Ottawa Hospital | Ottawa | ON | Type II | Medical |
| 5-Feb-16 | The Ottawa Hospital | Ottawa | ON | Type II | Medical |
| 5-Feb-16 | The Ottawa Hospital | Ottawa | ON | Type II | Medical |
| 5-Feb-16 | The Ottawa Hospital | Ottawa | ON | Type II | Medical |
| 5-Feb-16 | The Ottawa Hospital | Ottawa | ON | Type II | Medical |
| 5-Feb-16 | The Ottawa Hospital | Ottawa | ON | Type II | Medical |
| 5-Feb-16 | The Ottawa Hospital | Ottawa | ON | Type II | Medical |
| 5-Feb-16 | University Health Network | Toronto | ON | Type II | Commercial |
| 8-Feb-16 | 1788966 Alberta Ltd. | Redcliff | AB | Type II | Industrial |
| 8-Feb-16 | IKO Industries Ltd. | Brampton | ON | Type II | Industrial |
| 8-Feb-16 | Les Laboratoires d'Essais Mequaltech Inc. | Montréal | QC | Type II | Industrial |
| 8-Feb-16 | Mistras Services Inc. | Lévis | QC | Type II | Industrial |
| 8-Feb-16 | Seymour Pacific Developments Ltd. | Regina | SK | Type II | Industrial |
| 8-Feb-16 | Southern Alberta Institute of Technology | Calgary | AB | Type II | Industrial |
| 8-Feb-16 | Steel Inspection & Testing Ltd. | St. Catherines | ON | Type II | Industrial |
| 8-Feb-16 | Tarkett Inc. | Farnham | QC | Type II | Industrial |
| 9-Feb-16 | Canadian Institute for NDE | Hamilton | ON | Type II | Industrial |
| 9-Feb-16 | Englobe Corp. | Laval | QC | Type II | Industrial |
| 9-Feb-16 | Les laboratoires d'Essais Mequaltech Inc. | Montréal | QC | Type II | Industrial |
| 9-Feb-16 | Trenergy Inc. | St. Catherines | ON | Type II | Industrial |
| 9-Feb-16 | Vancouver Coastal Health Authority | Vancouver | BC | Type II | Medical |
| 10-Feb-16 | Cascades Canada ULC | Kingsey Falls | QC | Type II | Industrial |
| 10-Feb-16 | Centre hospitalier Ste-Croix | Drummondville | QC | Type II | Medical |
| 10-Feb-16 | Centre hospitalier Ste-Croix | Drummondville | QC | Type II | Medical |
| 11-Feb-16 | CHU de Québec – Université Laval | Sainte-Foy | QC | Type II | Medical |
| 11-Feb-16 | CHU de Québec – Université Laval | Sainte-Foy | QC | Type II | Medical |
| 11-Feb-16 | CHU de Québec – Université Laval | Ste-Foy | QC | Type II | Medical |
| 11-Feb-16 | CHU de Québec – Université Laval | Ste-Foy | QC | Type II | Medical |
| 11-Feb-16 | Di-Med Services Ltd. | Sainte-Foy | QC | Type II | Medical |
| 11-Feb-16 | Englobe Corp. | Laval | QC | Type II | Industrial |
| 11-Feb-16 | Nuclear Services Canada Inc. | Merlin | ON | Type II | Commercial |
| 11-Feb-16 | Unique Detection Services Ltd. | Cambridge | ON | Type II | Industrial |
| 11-Feb-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| 12-Feb-16 | CHU de Québec – Université Laval | Sainte-Foy | QC | Type II | Medical |
| 12-Feb-16 | CHU de Québec – Université Laval | Sainte-Foy | QC | Type II | Medical |
| 12-Feb-16 | CHU de Québec – Université Laval | Sainte-Foy | QC | Type II | Medical |
| 12-Feb-16 | CHU de Québec – Université Laval | Ste-Foy | QC | Type II | Medical |
| 12-Feb-16 | CHU de Québec – Université Laval | Ste-Foy | QC | Type II | Medical |
| 12-Feb-16 | CHU de Québec – Université Laval | Ste-Foy | QC | Type II | Medical |
| 12-Feb-16 | CHU de Québec – Université Laval | Ste-Foy | QC | Type II | Medical |
| 12-Feb-16 | GeoTerre Ltd. | Brampton | ON | Type II | Industrial |
| 15-Feb-16 | GeoNorth Engineering Ltd. | Prince George | BC | Type II | Industrial |
| 15-Feb-16 | St-Isodore Aspahalte Ltée | St-Isodore | NB | Type II | Industrial |
| 16-Feb-16 | 1583023 Alberta Ltd. | St. Albert | AB | Type II | Industrial |
| 16-Feb-16 | Beauty Packaging Canada, Inc. | Brampton | ON | Type II | Industrial |
| 16-Feb-16 | McElhanney Consulting Services Ltd. | Courtenay | BC | Type II | Industrial |
| 16-Feb-16 | Mills Memorial Hospital | Terrace | BC | Type II | Medical |
| 16-Feb-16 | Mills Memorial Hospital | Terrace | BC | Type II | Medical |
| 16-Feb-16 | NDC Infrared Engineering, Inc. | Irwnindale | CA | Type II | Commercial |
| 16-Feb-16 | Ontario Power Generation Inc. | Whitby | ON | Type II | Industrial |
| 16-Feb-16 | RTD Quality Services Inc. | Burlington | ON | Type II | Industrial |
| 16-Feb-16 | St. Michael's Hospital | Toronto | ON | Type II | Academic & research |
| 16-Feb-16 | St. Michael's Hospital | Toronto | ON | Type II | Academic & research |
| 16-Feb-16 | St. Michael's Hospital | Toronto | ON | Type II | Medical |
| 16-Feb-16 | St. Michael's Hospital | Toronto | ON | Type II | Medical |
| 16-Feb-16 | St. Michael's Hospital | Toronto | ON | Type II | Medical |
| 16-Feb-16 | St. Michael's Hospital | Toronto | ON | Type II | Medical |
| 16-Feb-16 | Trenergy Inc. | St. Catherines | ON | Type II | Industrial |
| 16-Feb-16 | Trevali Mining (New Brunswick) Ltd. | Bathurst | NB | Type II | Industrial |
| 17-Feb-16 | Agrium | Fort Saskatchewan | AB | Type II | Industrial |
| 17-Feb-16 | Canada Border Services Agency | Ottawa | ON | Type II | Industrial |
| 17-Feb-16 | Civil ArSa Engineering Inc. | Innisfil | ON | Type II | Industrial |
| 17-Feb-16 | Coco Paving (1990) Inc. | North York | ON | Type II | Industrial |
| 17-Feb-16 | Héma-Québec | Saint-Laurent | QC | Type II | Medical |
| 17-Feb-16 | Le Groupe Roy Consultants Ltée. | Bathurst | NB | Type II | Industrial |
| 17-Feb-16 | Peto MacCallum Ltd. | Kitchener | ON | Type II | Industrial |
| 17-Feb-16 | PML Inspection Services Ltd. | Fort Saskatchewan | AB | Type II | Industrial |
| 17-Feb-16 | Stantec Consulting Ltd. | Barrie | ON | Type II | Industrial |
| 18-Feb-16 | Aim Recycling Bathurst | Bathurst | NB | Type II | Industrial |
| 18-Feb-16 | Delwisch Developments Ltd. | Smithers | BC | Type II | Industrial |
| 18-Feb-16 | Ground Engineering & Materials Consultants Ltd. | Fredericton | NB | Type II | Industrial |
| 18-Feb-16 | Ground Engineering & Materials Consultants Ltd. | Fredericton | NB | Type II | Industrial |
| 18-Feb-16 | McElhanney Consulting Services Ltd. | Courtenay | BC | Type II | Industrial |
| 18-Feb-16 | Ontario Power Generation Inc. | Whitby | ON | Type II | Industrial |
| 18-Feb-16 | Régie régionale de la santé A | Bathurst | NB | Type II | Medical |
| 18-Feb-16 | Regional Health Authority A | Bathurst | NB | Type II | Medical |
| 18-Feb-16 | Sarafinchin Associates Ltd. | Toronto | ON | Type II | Industrial |
| 18-Feb-16 | University of Alberta | Edmonton | AB | Type II | Commercial |
| 19-Feb-16 | Acuren Group Inc. | Edmonton | AB | Type II | Industrial |
| 19-Feb-16 | Medical Imaging Consultants | Edmonton | AB | Type II | Medical |
| 19-Feb-16 | Riverview Animal Hospital | Riverview | NB | Type II | Medical |
| 22-Feb-16 | Englobe Corp. | Laval | QC | Type II | Industrial |
| 22-Feb-16 | Labo S.M. Inc. | Longeuil | QC | Type II | Industrial |
| 22-Feb-16 | Les Tricots Duval & Raymond Ltée | Princeville | QC | Type II | Industrial |
| 22-Feb-16 | Maskimo Construction Inc. | Trois-Rivières | QC | Type II | Industrial |
| 23-Feb-16 | 1388020 Ontario Corp. | Toronto | ON | Type II | Medical |
| 23-Feb-16 | Acuren Group Inc. | Edmonton | AB | Type II | Industrial |
| 23-Feb-16 | Bonnett's Energy Services Ltd. | Red Deer | AB | Type II | Industrial |
| 23-Feb-16 | Cégep Limoilou | Québec | QC | Type II | Industrial |
| 23-Feb-16 | Centre de santé et de services sociaux D'Arthabaska-Érable | Victoriaville | QC | Type II | Medical |
| 23-Feb-16 | Centre de santé et de services sociaux D'Arthabaska-Érable | Victoriaville | QC | Type II | Medical |
| 23-Feb-16 | GHD Consultants Ltd. | Saint-Laurent | QC | Type II | Industrial |
| 23-Feb-16 | GHD Consultants Ltd. | Saint-Laurent | QC | Type II | Industrial |
| 23-Feb-16 | Humber River Hospital | Toronto | ON | Type II | Medical |
| 23-Feb-16 | Humber River Hospital | Toronto | ON | Type II | Medical |
| 23-Feb-16 | Inventiv Health Clinique Inc. | Québec | QC | Type II | Academic & research |
| 23-Feb-16 | Louis W. Bray Construction Ltd. | St. Andrews West | ON | Type II | Industrial |
| 23-Feb-16 | Loyalist College of Applied Arts and Technology | Belleville | ON | Type II | Industrial |
| 23-Feb-16 | Vétoquinol North America Inc. | Belleville | ON | Type II | Academic & research |
| 24-Feb-16 | Avizo Experts-Conseils Inc. | Sherbrooke | QC | Type II | Industrial |
| 24-Feb-16 | Blue Mountain Wallcoverings Inc. | Sherbrooke | QC | Type II | Industrial |
| 24-Feb-16 | Certified Testing Systems (2009) Inc. | Kitchener | ON | Type II | Industrial |
| 24-Feb-16 | EXP Services Inc. | Laval | QC | Type II | Industrial |
| 24-Feb-16 | Honeywell Ltd./Honeywell Ltée | Mississauga | ON | Type II | Commercial |
| 24-Feb-16 | Institut universitaire en santé mentale de Québec | Québec | QC | Type II | Academic & research |
| 24-Feb-16 | SGS Canada Ltd. | Lakefield | ON | Type II | Industrial |
| 24-Feb-16 | SGS Canada Ltd. | Lakefield | ON | Type II | Industrial |
| 25-Feb-16 | Construction DJL Inc. | Boucherville | QC | Type II | Industrial |
| 25-Feb-16 | lantheus MI Canada, Inc. | Montréal | QC | Type II | Commercial |
| 25-Feb-16 | Les Laboratoires d'Essais Mequaltech Inc. | Montréal | QC | Type II | Industrial |
| 25-Feb-16 | Sintra Inc. | Montréal | QC | Type II | Industrial |
| 26-Feb-16 | Amec Foster Wheeler Americas Ltd./Amec Foster Wheeler Améeriques Ltée | Lloydminster | AB | Type II | Industrial |
| 26-Feb-16 | Construction Testing Asphalt Lab Ltd. | Cambridge | ON | Type II | Industrial |
| 26-Feb-16 | Isologic Innovative Radiopharmaceuticals of Ontario Ltd. | Ottawa | ON | Type II | Commercial |
| 26-Feb-16 | Isologic Innovative Radiopharmaceuticals of Ontario Ltd. | Ottawa | ON | Type II | Commercial |
| 29-Feb-16 | Canadian Nuclear Laboratories Ltd. | Chalk River | ON | Type II | Academic & research |
| 29-Feb-16 | Canadian Nuclear Laboratories Ltd. | Chalk River | ON | Type II | Academic & research |
| 29-Feb-16 | Centre Hospitalier Universitaire de Québec | Québec | QC | Type II | Academic & research |
| 29-Feb-16 | R-Metrics | Burlington | ON | Type II | Commercial |
| Inspection date | Licensee name | City | Province | Inpection type | Sector |
|---|---|---|---|---|---|
| 1-Mar-16 | Perfection Inspection Ltd. | Cambridge | ON | Type II | Industrial |
| 1-Mar-16 | Perfection Inspection Ltd. | Cambridge | ON | Type II | Industrial |
| 1-Mar-16 | Université Laval | Québec | QC | Type II | Academic & research |
| 1-Mar-16 | Windsor Regional Hospital | Windsor | ON | Type II | Medical |
| 2-Mar-16 | Certified Testing Systems (2009) Inc | Kitchener | ON | Type II | Industrial |
| 2-Mar-16 | Certified Testing Systems (2009) Inc | Kitchener | ON | Type II | Industrial |
| 3-Mar-16 | Acuren Group Inc. | Brossard | QC | Type II | Industrial |
| 3-Mar-16 | Big Guns Energy Services Inc. | Calgary | AB | Type II | Industrial |
| 3-Mar-16 | Fisher Scientific Company | Ottawa | ON | Type II | Commercial |
| 3-Mar-16 | McGill University Health Centre | Montréal | QC | Type II | Medical |
| 3-Mar-16 | Nuclear Services Canada Inc | Merlin | ON | Type II | Commercial |
| 3-Mar-16 | Voltage Wireline Inc. | Blackfalds | AB | Type II | Industrial |
| 4-Mar-16 | General Dynamics Produits de defense et Systemes Tactiques – Canada Inc. | Le Gardeur | QC | Type II | Industrial |
| 7-Mar-16 | Arbutus Biopharma Corporation | Burnaby | BC | Type II | Academic & research |
| 8-Mar-16 | A & A Concrete X-Ray and Coring Ltd. | Langley | BC | Type II | Industrial |
| 8-Mar-16 | Custom Fabricators & Machinists Limited / Fabricants et Mach | Saint John | NB | Type II | Industrial |
| 8-Mar-16 | Custom Fabricators & Machinists Limited / Fabricants et Mach | Saint John | NB | Type II | Industrial |
| 8-Mar-16 | Honeywell Ltd. | Lachine | QC | Type II | Commercial |
| 8-Mar-16 | Jubilant DraxImage Inc. | Kirkland | QC | Type II | Commercial |
| 8-Mar-16 | Lafarge Canada Inc. | Richmond | BC | Type II | Industrial |
| 8-Mar-16 | Law Engineering (London) Inc. | London | ON | Type II | Industrial |
| 8-Mar-16 | RTD Quality Services Inc. | Burlington | ON | Type II | Industrial |
| 8-Mar-16 | RTD Quality Services Inc. | Burlington | ON | Type II | Industrial |
| 8-Mar-16 | St. Joseph's Health Care, London | London | ON | Type II | Medical |
| 8-Mar-16 | Taghleef Industries Canada Inc. | Varennes | QC | Type II | industrial |
| 9-Mar-16 | Acuren Group Inc. | Edmonton | AB | Type II | Industrial |
| 9-Mar-16 | Acuren Group Inc. | Edmonton | AB | Type II | Industrial |
| 9-Mar-16 | Coco Paving (1990) Inc. | North York | ON | Type II | Industrial |
| 9-Mar-16 | Gerdau AmeriSteel Corporation | Whitby | ON | Type II | Industrial |
| 9-Mar-16 | Les Laboratoires d'Essais Mequaltech Inc. | Montréal | QC | Type II | Industrial |
| 9-Mar-16 | Vancouver Coastal Health Authority | Vancouver | BC | Type II | Medical |
| 10-Mar-16 | Acuren Group Inc. | Edmonton | AB | Type II | Industrial |
| 10-Jan-16 | Cargill Limited | Clavet | SK | Type II | Industrial |
| 10-Mar-16 | Englobe Corp. | Laval | QC | Type II | Industrial |
| 10-Mar-16 | Government of Newfoundland & Labrador | St. John's | NL | Type II | Industrial |
| 10-Mar-16 | PML Inspection Services Ltd. | Fort Saskatchewan | AB | Type II | Industrial |
| 10-Mar-16 | Premier Diagnostic Center (Vancouver) Inc. | Burnaby | BC | Type II | Medical |
| 11-Mar-16 | Les Laboratoires d'Essais Mequaltech Inc. | Montréal | QC | Type II | Industrial |
| 11-Mar-16 | Tomlinson Enterprises Ltd. | Sarnia | ON | Type II | Industrial |
| 14-Mar-16 | Baker Hughes Canada Company | Leduc | AB | Type II | Industrial |
| 14-Mar-16 | Baker Hughes Canada Company | Leduc | AB | Type II | Industrial |
| 14-Mar-16 | Bot Construction Limited | Oakville | ON | Type II | Industrial |
| 14-Mar-16 | Coca-Cola Refreshments Canada Company | Brampton | ON | Type II | Industrial |
| 14-Mar-16 | MyHealth Partners Inc. | Toronto | ON | Type II | Medical |
| 14-Mar-16 | Orbit Engineering Limited | Brampton | ON | Type II | Industrial |
| 14-Mar-16 | Peto MacCallum Ltd. | Kitchener | ON | Type II | Industrial |
| 14-Mar-16 | Toronto Cardiology Associates Inc | Toronto | ON | Type II | Medical |
| 15-Mar-16 | Canadian Inspection Ltd. | Edmonton | AB | Type II | Industrial |
| 15-Mar-16 | Pine Environmental Services Inc. | Mississauga | ON | Type II | Commercial |
| 16-Mar-16 | CEGEP de Trois-Rivières | Trois-Rivière | QC | Type II | Industrial |
| 16-Mar-16 | Labatt Brewing Company Ltd. / La Brasserie Labatt limitée | London | ON | Type II | Industrial |
| 17-Mar-16 | Woodstock General Hospital | Woodstock | ON | Type II | Medical |
| 18-Mar-16 | Nuclear Services Canada Inc. | Merlin | ON | Type II | Commercial |
| 21-Mar-16 | Hamilton Health Sciences Corporation/St. Joseph's Health System | Hamilton | ON | Type II | Medical |
| 22-Mar-16 | Acuren Inc. | Edmonton | AB | Type II | Industrial |
| 22-Mar-16 | Atlantic Coated Papers Ltd. / Papiers Couchés d'Atlantic Lté | Windsor | QC | Type II | Industrial |
| 22-Mar-16 | Couillard Construction Limitée | Coaticook | QC | Type II | Industrial |
| 22-Mar-16 | Hamilton Health Sciences Corp. | Hamilton | ON | Type II | Medical |
| 22-Mar-16 | Hamilton Health Sciences Corp. | Hamilton | ON | Type II | Medical |
| 22-Mar-16 | Qualité N.D.E. Limitée | Mercier | QC | Type II | Commercial |
| 23-Mar-16 | Mistras Services Inc. | Lévis | QC | Type II | Industrial |
| 23-Mar-16 | The Corporation of the City of Oshawa | Oshawa | ON | Type II | Industrial |
| 23-Mar-16 | The Graff Company | Mississauga | ON | Type II | Industrial |
| 23-Mar-16 | The Hospital for Sick Children | Toronto | ONC | Type II | Academic & research |
| 24-Mar-16 | Canadian Tower Scanning Inc. | Sarnia | ON | Type II | Industrial |
| 29-Mar-16 | BWXT Canada Ltd. | Cambridge | ON | Type II | Industrial |
| 29-Mar-16 | Cepsa Chimie Bécancour Inc. / Cepsa Química Bécancour Inc. | Bécancour | QC | Type II | Industrial |
| 29-Mar-16 | Unique Detection Services Ltd. | Cambridge | ON | Type II | Industrial |
| 30-Mar-16 | Christopher Thompson | Montréal | QC | Type II | Commercial |
| 31-Mar-16 | Halton HealthCare Services Corporation | Oakville | ON | Type II | Medical |
| 31-Mar-16 | Halton HealthCare Services Corporation | Oakville | ON | Type II | Medical |
| 31-Mar-16 | Kodiak Quality Control Ltd. | Oakville | ON | Type II | Commercial |
| 5-Apr-16 | Tomlinson Enterprises Ltd. | Sarnia | ON | Type II | Industrial |
| 6-Apr-16 | Environment Canada | Burlington | ON | Type II | Academic & research |
| 6-Apr-16 | GHD Consultants Ltd. | Saint-Laurent | QC | Type II | Industrial |
| 6-Apr-16 | Stern Laboratories Inc. | Hamilton | ON | Type II | Academic & research |
| 7-Apr-16 | 1051630 Alberta Ltd | Calgary | AB | Type II | Industrial |
| 7-Apr-16 | Canadian Food Inspection Agency | Guelph | ON | Type II | Academic & research |
| 7-Apr-16 | Englobe Corp | Laval | QC | Type II | Industrial |
| 7-Apr-16 | MPE Engineering Ltd | Lethbridge | AB | Type II | Industrial |
| 7-Apr-16 | Weatherford Canada Ltd. | Calgary | AB | Type II | Industrial |
| 8-Apr-16 | 1051630 Alberta Ltd. | Calgary | AB | Type II | Industrial |
| 8-Apr-16 | G Tech Earth Sciences Corp | Magrath | AB | Type I | Industrial |
| 8-Apr-16 | Lethbridge College | Lethbridge | ON | Type II | Industrial |
| 12-Apr-16 | Amec Foster Wheeler Americas Limited / Amec Foster Wheeler Amériques Limitée | Lloydminster | AB | Type II | Industrial |
| 12-Apr-16 | Amec Foster Wheeler Americas Limited / Amec Foster Wheeler Amériques Limitée | Lloydminster | AB | Type II | Medical |
| 12-Apr-16 | McIntosh Lalani Engineering Ltd. | Calgary | AB | Type II | Industrial |
| 12-Apr-16 | McIntosh Lalani Engineering Ltd. | Calgary | AB | Type II | Industrial |
| 13-Apr-16 | Curtis Engineering Associates Ltd. | Calgary | AB | Type II | Industrial |
| 13-Apr-16 | Curtis Engineering Associates Ltd. | Calgary | AB | Type II | Industrial |
| 13-Apr-16 | Global Engineering & Testing Ltd | Calgary | AB | Type II | Industrial |
| 13-Apr-16 | Golder Associates Ltd. | Mississauga | ON | Type II | Industrial |
| 14-Apr-16 | Amec Foster Wheeler Americas Limited / Amec Foster Wheeler Amériques Limitée | Lloydminster | AB | Type I | Industrial |
| 18-Apr-16 | Frontop Engineering Limited | Markham | ON | Type II | Industrial |
| 18-Apr-16 | Golder Associates Ltd. | Mississauga | ON | Type II | Industrial |
| 25-Apr-16 | 1051630 Alberta Ltd | Calgary | AB | Type II | Industrial |
| 26-Apr-16 | Coco Paving (1990) Inc. | Tecumseh | ON | Type II | Industrial |
| 26-Apr-16 | Davroc Testing Laboratories Inc. | Brammpton | ON | Type II | Indutstrial |
| 26-Apr-16 | Davroc Testing Laboratories Inc. | Brampton | ON | Type II | Industrial |
| 26-Apr-16 | GHD Consultants Ltd. | Montréal | QC | Type II | Industrial |
| 27-Apr-16 | J.L. Shepherd and Associates | San Fernando | CA | Type II | Commercial |
| 27-Apr-16 | Okanagan-Kootenay | Osoyoos | BC | Type II | Medical |
| 27-Apr-16 | Regional Municipality of Durham | Whitby | ON | Type II | Industrial |
| 27-Apr-16 | TISI Canada Inc. | Oakville | ON | Type II | Industrial |
| 28-Apr-16 | AEP Canada Inc. | West Hill | ON | Type II | Industrial |
| 28-Apr-16 | Ecora Engineering Ltd. | Kelowna | BC | Type II | Industrial |
| 28-Apr-16 | Kidde Canada Inc. | Vaughan | ON | Type II | Commercial |
| 28-Apr-16 | Klöckner Pentaplast Company | Montréal | QC | Type II | Industrial |
| 28-Apr-16 | Lakeridge Health | Oshawa | ON | Type II | Medical |
| 28-Apr-16 | Lakeridge Health | Oshawa | ON | Type II | Medical |
| 28-Apr-16 | Omnifission Inc. | Brampton | ON | Type II | Commercial |
| 28-Apr-16 | Université Concordia/ Concordia University | Montréal | ON | Type II | Academic & research |
| 29-Apr-16 | EXL Engineering Inc. | Delta | BC | Type II | Indutrial |
| 29-Jan-16 | EXP Services Inc. | Brampton | ON | Type II | Industrial |
| 29-Apr-16 | Fletcher Paine Associates Ltd. | Vernon | BC | Type II | Industrial |
| 29-Apr-16 | Hunt Inspection Ltd | Stettler | AB | Type II | Industrial |
| 29-Apr-16 | Insite Materials Testing Group Ltd. | Kelowna | BC | Type II | Industrial |
| 29-Apr-16 | Interior Testing Services Ltd. | Kelowna | BC | Type II | Industrial |
| Inspection date | Licensee name | City | Province | Inpection type | Sector |
|---|---|---|---|---|---|
| 2-May-16 | Celanese Eva Performance Polymers Inc. | Edmonton | AB | Type II | Industrial |
| 3-May-16 | Acuren Group Inc. | Edmonton | AB | Type II | Industrial |
| 3-May-16 | Centre universitaire de santé McGill / McGill University Health Centre | Montréal | QC | Type II | Medical |
| 3-May-16 | City of Edmonton, Engineering Services Section | Edmonton | AB | Type II | Industrial |
| 3-May-16 | Golder Associates Ltd. | Mississauga | ON | Type II | Industrial |
| 3-May-16 | Opus Stewart Weir Ltd. | Sherwood Park | AB | Type II | Industrial |
| 3-May-16 | ShawCor Ltd. | Toronto | ON | Type II | Industrial |
| 3-May-16 | Stuart Hunt & Associates Ltd. | St. Albert | AB | Type II | Commercial |
| 3-May-16 | Syncrude Canada Ltd. | Edmonton | AB | Type II | Industrial |
| 3-May-16 | Syncrude Canada Ltd. | Edmonton | AB | Type II | Academic & research |
| 4-May-16 | Centre universitaire de santé McGill / McGill University Health Centre | Montréal | QC | Type II | Medical |
| 4-May-16 | Centre universitaire de santé McGill / McGill University Health Centre | Montréal | QC | Type II | Medical |
| 4-May-16 | Centre universitaire de santé McGill / McGill University Health Centre | Montréal | QC | Type II | Medical |
| 4-May-16 | PCL Construction Management Inc. | Edmonton | AB | Type II | Industrial |
| 4-May-16 | Ready Engineering Corporation | Spruce Grove | AB | Type II | Commercial |
| 4-May-16 | Thermo EGS Gauging, Inc. | Wilmington | MA | Type II | Commercial |
| 4-May-16 | Thurber Engineering Ltd. | Calgary | AB | Type II | Industrial |
| 4-May-16 | Tuboscope Vetco Canada ULC | Nisku | AB | Type II | Industrial |
| 5-May-16 | British Columbia Cancer Agency | Surrey | BC | Type II | Medical |
| 5-May-16 | Clifton Associates Ltd. | Regina | SK | Type II | Industrial |
| 5-May-16 | Omnifission Inc. | Brampton | ON | Type II | Commercial |
| 5-May-16 | Regie regional de la santé A | Moncton | NB | Type II | Medical |
| 5-May-16 | Regie regional de la santé A | Moncton | NB | Type II | Medical |
| 5-May-16 | Regie regional de la santé A | Moncton | NB | Type II | Medical |
| 5-May-16 | Rivest Technologies Incorporated | Edmonton | AB | Type II | Industrial |
| 5-May-16 | Services New Brunswick | Fredricton | NB | Type II | Commercial |
| 5-May-16 | Trillium Beverage Inc. | North York | ON | Type II | Industrial |
| 5-May-16 | Windsor Regional Hospital | Windsor | ON | Type II | Medical |
| 5-May-16 | Windsor Regional Hospital | Windsor | ON | Type II | Medical |
| 6-May-16 | Canada Pump and Power (CPP) Corporation | Ardrossan | AB | Type II | Industrial |
| 6-May-16 | Centre universitaire de santé McGill / McGill University Health Centre | Montréal | QC | Type II | Medical |
| 6-May-16 | Centre universitaire de santé McGill / McGill University Health Centre | Montréal | QC | Type II | Medical |
| 6-May-16 | Metro Testing Laboratories Ltd. | Burnaby | BC | Type II | Industrial |
| 9-May-16 | Domtar Inc. | Dyden | ON | Type II | Industrial |
| 9-May-16 | Kam Tech Quality Management Inc. | Kamloops | BC | Type II | Industrial |
| 10-May-16 | Highland Valley Copper | Logan Lake | BC | Type II | Industrial |
| 11-May-16 | 8418748 Canada Inc. | Montréal | QC | Type II | Industrial |
| 11-May-16 | Coveris Flexibles Canada Inc. | Whitby | ON | Type II | Industrial |
| 11-May-16 | Global Engineering & Testing Ltd | Calgary | AB | Type II | Industrial |
| 11-May-16 | Rouge Valley Health System | Scarborough | ON | Type II | Medical |
| 11-May-16 | Trans Mountain Pipeline ULC | Sherwood Park | AB | Type II | Industrial |
| 11-May-16 | Trans Mountain Pipeline ULC | Sherwod Park | AB | Type II | Industrial |
| 12-May-16 | Amec Foster Wheeler Americas Limited / Amec Foster Wheeler A | Lloydminster | AB | Type II | Industrial |
| 12-May-16 | EXP Services Inc. | Brampton | ON | Type II | Industrial |
| 12-May-16 | Kam Tech Consulting Incorporated | Kamloops | BC | Type II | Industrial |
| 12-May-16 | Ontario Power Generation Inc. | Bowmanville | ON | Type II | Academic & research |
| 12-May-16 | SGS Canada Inc. | Lakefield | ON | Type II | Industrial |
| 13-May-16 | EXP Services Inc. | Brampton | ON | Type II | Industrial |
| 13-May-16 | EXP Services Inc. | Brampton | ON | Type II | Industrial |
| 13-May-16 | Terraprobe Testing Ltd. | Brampton | ON | Type II | Industrial |
| 13-May-16 | Terraprobe Testing Ltd. | Brampton | ON | Type II | Industrial |
| 16-May-16 | 3M Canada Company | London | ON | Type II | Industrial |
| 16-May-16 | EXP Services Inc. | Brampton | ON | Type II | Industrial |
| 16-May-16 | Fortress Specialty Cellulose Inc. | Thurso | QC | Type II | Industrail |
| 16-May-16 | Groupe ABS Inc. | St-Rémi | QC | Type II | Industrial |
| 16-May-16 | KPGP Inc. | Gatineau | QC | Type II | Industrial |
| 16-May-16 | M.J. Davenport & Associates Ltd | Peterborough | ON | Type II | Industrial |
| 16-May-16 | Trent University | Peterborough | ON | Type II | Academic & research |
| 17-May-16 | Advanced Gauging Technologies, L.L.C. | Plain City | OH | Type II | Commercial |
| 17-May-16 | Amec Foster Wheeler Americas Limited / Amec Foster Wheeler A | Lloydminster | AB | Type II | Industrial |
| 17-Jan-16 | Berry Plastics Canada Inc. | Belleville | ON | Type II | Industrial |
| 17-May-16 | Cascades Canada ULC | Kingsey Falls | QC | Type II | Industrial |
| 17-May-16 | Commandité Papier Masson WB Ltée | Gatineau | QC | Type II | Industrial |
| 17-May-16 | Construction Control Inc. | Concord | ON | Type II | Industrial |
| 17-May-16 | Construction DJL Inc. | Boucherville | QC | Type II | Industrial |
| 17-May-16 | Englobe Corp. | Laval | QC | Type II | Industrial |
| 17-May-16 | GHD Consultants Ltd. | Montréal | QC | Type II | Industrial |
| 17-May-16 | Groupe ABS Inc | St-Rémi | QC | Type II | Industrial |
| 17-May-16 | Samuel, Son & Co. Limited | Stoney Creek | ON | Type II | Industrial |
| 17-May-16 | Sonoco Canada Corporation | Trenton | ON | Type II | Industrial |
| 17-May-16 | UTC Fire & Security Canada Inc. operating as Chubb Edwards | Mississauga | ON | Type II | Commercial |
| 17-May-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| 18-May-16 | Centre de santé et de services sociaux de Gatineau | Gatineau | QC | Type II | Medical |
| 18-May-16 | Centre de santé et de services sociaux de Gatineau | Gatineau | QC | Type II | Medical |
| 18-May-16 | Coco Paving (1990) Inc. | Tecumseh | ON | Type II | Industrial |
| 18-May-16 | Essroc Canada Inc. | Picton | ON | Type II | Industrial |
| 18-May-16 | General Dynamics Land Systems – Canada Corp. | London | ON | Type II | Commercial |
| 18-May-16 | General Dynamics Land Systems – Canada Corp. | London | ON | Type II | Commercial |
| 18-May-16 | General Dynamics Land Systems – Canada Corp. | London | ON | Type II | Commercial |
| 18-May-16 | General Dynamics Land Systems – Canada Corp. | London | ON | Type II | Commercial |
| 18-May-16 | GHD Consultants Ltd. | Montréal | QC | Type II | Industrial |
| 18-May-16 | J & P Levesque Bros Haulage Ltd. | Bancroft | ON | Type II | Industrial |
| 18-May-16 | SNC-Lavalin GEM Québec Inc. | Montréal | QC | Type I | Industrial |
| 19-May-16 | Labatt Brewing Company Ltd. / La Brasserie Labatt limitée | London | ON | Type II | Industrial |
| 19-May-16 | MyHealth Partners Inc. | Toronto | ON | Type II | Medical |
| 19-May-16 | Thomas Cavanagh Construction Limitedl | Ashton | ON | Type II | Industrial |
| 20-May-16 | Golder Associates Ltd. | Mississauga | ON | Type II | Industrial |
| 20-May-16 | Medical Imaging Consultants | Edmonton | AB | Type II | Medical |
| 20-May-16 | Philips Electronics Ltd./Philips Electronique Ltée | Markham | ON | Type II | Commercial |
| 20-May-16 | Resolute FP Canada Inc. / PF Résolu Canada Inc. | Gatineau | QC | Type II | Industrial |
| 20-May-16 | Stratford General Hospital | Stratford | ON | Type II | Medical |
| 20-May-16 | Stratford General Hospital | Stratford | ON | Type II | Medical |
| 24-May-16 | Englobe Corp. | Montréal | QC | Type I | Industrial |
| 24-Jan-16 | Thurber Engineering Ltd. | Calgary | AB | Type II | Industrial |
| 25-May-16 | Aecom Canada Ltd. | Edmonton | AB | Type II | Industrial |
| 25-May-16 | Cytec Canada Inc. | Niagara Falls | ON | Type II | Industrial |
| 25-May-16 | Endress + Hauser Canada Ltd./ Endress + Hauser Canada Ltée | Burlington | ON | Type II | Commercial |
| 25-May-16 | Golder Associates Ltd. | Mississauga | ON | Type II | Industrial |
| 25-May-16 | Golder Associates Ltd. | Mississauga | ON | Type II | Industrial |
| 25-May-16 | Golder Associates Ltd. | Saskatoon | SK | Type II | Academic & research |
| 25-May-16 | P. Machibroda Engineering Ltd. | Saskatoon | SK | Type II | Industrial |
| 25-May-16 | PanPacific Wireline Services Inc. | Brossard | QC | Type II | Industrial |
| 25-May-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| 25-May-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| 26-May-16 | ABB Inc. | St-Laurent | QC | Type II | Commercial |
| 26-May-16 | ABB Inc. | St-Laurent | QC | Type II | Commercial |
| 26-May-16 | Allnorth Consultants Limited | Terrace | BC | Type II | Industrial |
| 26-May-16 | Amec Foster Wheeler Americas Limited / Amec Foster Wheeler Amériques Ltée | Lloydminster | AB | Type II | Industrial |
| 26-May-16 | North West Nuclear Medicine for Animals Inc. | Vancouver | BC | Type II | Medical |
| 27-May-16 | Associated Engineering (Sask.) Ltd. | Saskatoon | SK | Type II | Industrial |
| 27-May-16 | SNC-Lavalin Inc. | Saskatoon | SK | Type II | Industrial |
| 27-May-16 | SNC-Lavalin Inc. | Saskatoon | SK | Type II | Academic & research |
| 30-May-16 | Kresin Engineering Corporation | Sault Ste. Marie | ON | Type II | Industrial |
| 30-May-16 | Lac des Iles Mines Ltd. | Thunder Bay | ON | Type II | Industrial |
| 30-May-16 | Pioneer Construction Inc. | Copper Cliff | ON | Type II | Industrial |
| 31-May-16 | Acuren Inc. | Edmonton | AB | Type II | Industrial |
| 31-May-16 | Englobe Corp. | Laval | QC | Type II | Industrial |
| 31-May-16 | EXP Services Inc. | Laval | QC | Type II | Industrial |
| 31-May-16 | Hatch Ltd. | Clarenville | NL | Type II | Industrial |
| 31-May-16 | Mike Abbot | Toronto | ON | Type II | Industrial |
| 31-May-16 | Mike Abbot (Aurico Metals Inc.) | Toronto | ON | Type II | Industrial |
| 31-May-16 | Resolute FP Canada Inc. / PF Résolu Canada Inc. | Thunder Bay | ON | Type II | Industrial |
| 31-May-16 | Robert S. Wilson | Sault Ste. Marie | ON | Type II | Industrial |
| 31-May-16 | TBT Engineering Limited | Thunder Bay | ON | Type II | Industrial |
| 31-May-16 | Teranorth Construction & Engineering Ltd. | Sudbury | ON | Type II | Industrial |
| 31-May-16 | Tulloch Contract Administration Inc. | Thessalon | ON | Type II | Industrial |
| 1-Jun-16 | Centre de santé et de services sociaux de Gatineau | Gatineau | QC | Type II | Medical |
| 1-Jun-16 | Centre de santé et de services sociaux de Gatineau | Gatineau | QC | Type II | Medical |
| 1-Jun-16 | Essar Steel Algoma Inc. | Sault Ste. Marie | ON | Type II | Industrial |
| 1-Jun-16 | Hoban Equipment Ltd. | Vernon | BC | Type II | Industrial |
| 1-Jun-16 | Natural Resources Canada/ Ressources naturelles Canada | Ottawa | ON | Type II | Academic & research |
| 2-Jun-16 | Bruno's Contracting (Thunder Bay) Ltd. | Thunder Bay | ON | Type II | Industrial |
| 2-Jun-16 | DST Consulting Engineers Inc. | Kenora | ON | Type II | Industrial |
| 2-Jun-16 | Hatch Ltd. | Clarenville | NL | Type II | Industrial |
| 2-Jun-16 | The University Hospital of Northern British Columbia | Prince George | BC | Type II | Medical |
| 3-Jun-16 | Regional Municipality of Durham | Whitby | ON | Type II | Industrial |
| 3-Jun-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| 6-Jun-16 | Cott Corporation | Mississauga | ON | Type II | Industrial |
| 7-Jun-16 | University of British Columbia | Vancouver | BC | Type II | Academic & research |
| 8-Jun-16 | Associate Veterinary Clinics (1981) Ltd. | Calgary | AB | Type II | Medical |
| 8-Jun-16 | Peto MacCallum Ltd. | Kitchener | ON | Type II | Industrial |
| 8-Jun-16 | Shad & Associates Inc. | Vaughan | ON | Type II | Industrial |
| 8-Jun-16 | University of the Fraser Valley | Abbotsford | BC | Type II | Medical |
| 9-Jun-16 | Amgen British Columbia Inc. | Burnaby | BC | Type II | Medical |
| 9-Jun-16 | Groupe ABS Inc. | St-Rémi | QC | Type II | Industrial |
| 10-Jun-16 | The University Hospital of Northern British Columbia | Prince George | BC | Type II | Medical |
| 12-Jun-16 | BCG Engineering Inc. | Halifax | NS | Type II | Industrial |
| 12-Jun-16 | Red Chris Development Company Ltd. | Dease Lake | BC | Type II | Industrial |
| 13-Jun-16 | Allnorth Consultants Limited | Terrace | BC | Type II | Industrial |
| 13-Jun-16 | MyHealth Partners Inc. | Toronto | ON | Type II | Medical |
| 13-Jun-16 | Opus Stewart Weir Ltd. | Sherwood Park | AB | Type II | Industrial |
| 13-Jun-16 | Seymour Pacific Developments Ltd. | Regina | SK | Type II | Industrial |
| 14-Jun-16 | Advance Testing Ltd. | Surrey | BC | Type II | Industrial |
| 14-Jun-16 | McConnell Brain Imaging Center | Montréal | QC | Type II | Academic & research |
| 14-Jun-16 | McConnell Brain Imaging Center | Montréal | QC | Type II | Medical |
| 14-Jun-16 | McConnell Brain Imaging Center | Montréal | QC | Type II | Medical |
| 14-Jun-16 | McElhanney Consulting Services Ltd. | Courtenay | BC | Type II | Industrial |
| 14-Jun-16 | Montreal Neurological Institute | Montréal | QC | Type II | Commercial |
| 14-Jun-16 | Teknoscan Systems Inc. | Vaughan | ON | Type II | Commercial |
| 14-Jun-16 | University of Manitoba | Winnpeg | MB | Type II | Industrial |
| 14-Jun-16 | University of Manitoba | Winnpeg | MB | Type II | Academic & research |
| 14-Jun-16 | University of Manitoba | Winnpeg | MB | Type II | Academic & research |
| 14-Jun-16 | University of Manitoba | Winnpeg | MB | Type II | Academic & research |
| 14-Jun-16 | University of Manitoba | Winnpeg | MB | Type II | Academic & research |
| 14-Jun-16 | University of Manitoba | Winnpeg | MB | Type II | Academic & research |
| 14-Jun-16 | University of Manitoba | Winnpeg | MB | Type II | Academic & research |
| 14-Jun-16 | University of Manitoba | Winnpeg | MB | Type II | Academic & research |
| 14-Jun-16 | University of Manitoba | Winnpeg | MB | Type II | Academic & research |
| 14-Jun-16 | University of Manitoba | Winnpeg | MB | Type II | Academic & research |
| 14-Jun-16 | University of Manitoba | Winnpeg | MB | Type II | Academic & research |
| 14-Jun-16 | University of Manitoba | Winnpeg | MB | Type II | Academic & research |
| 14-Jun-16 | University of Manitoba | Winnpeg | MB | Type II | Academic & research |
| 14-Jun-16 | University of Manitoba | Winnpeg | MB | Type II | Academic & research |
| 14-Jun-16 | University of Manitoba | Winnpeg | MB | Type II | Academic & research |
| 14-Jun-16 | University of Manitoba | Winnpeg | MB | Type II | Academic & research |
| 14-Jun-16 | University of Manitoba | Winnpeg | MB | Type II | Academic & research |
| 15-Jun-16 | Amec Foster Wheeler Americas Limited / Amec Foster Wheeler Amériques Limitée | Lloydminster | AB | Type II | Industrial |
| 15-Jun-16 | GeoNorth Engineering Ltd. | Prince George | BC | Type II | Industrial |
| 16-Jun-16 | EXP Services Inc. | Laval | QC | Type II | Industrial |
| 16-Jun-16 | Goldcorp Canada Ltd. | Houston | BC | Type II | Industrial |
| 16-Jun-16 | Golder Associates Ltd. | Mississauga | ON | Type II | Industrial |
| 16-Jun-16 | Huckleberry Mines Ltd. | Houston | BC | Type II | Industrial |
| 16-Jun-16 | Huckleberry Mines Ltd. | Houston | BC | Type II | Industrial |
| 16-Jun-16 | Manitoba Transportation and Government Services | West St. Paul | MB | Type II | Industrial |
| 16-Jun-16 | Manitoba Transportation and Government Services | West St. Paul | MB | Type II | Industrial |
| 16-Jun-16 | NU-B Inc. | St-Laurent | QC | Type II | Industrial |
| 16-Jun-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| 17-Jun-16 | Allnorth Consultants Limited | Terrace | BC | Type II | Industrial |
| 17-Jun-16 | Englobe Corp. | Laval | QC | Type II | Industrial |
| 17-Jun-16 | GHD Consultants Ltd. | Montréal | QC | Type II | Industrial |
| 17-Jun-16 | Groupe ABS Inc. | St-Rémi | QC | Type II | Industrial |
| 17-Jun-16 | Groupe CRH Canada Inc. / CRH Canada Group Inc. | Laval | QC | Type II | Industrial |
| 17-Jun-16 | Groupe TNT Inc. / TNT Group Inc. | Boisbriand | QC | Type II | Industrial |
| 17-Jun-16 | Siemens Molecular Imaging, Inc. | Knoxville | TN | Type II | Commercial |
| 17-Jun-16 | SNC-Lavalin GEM Québec Inc. | Montréal | QC | Type II | Industrial |
| 17-Jun-16 | West-Can Inspection Ltd. | Winnipeg | MB | Type II | Industrial |
| 20-Jun-16 | Groupe ABS Inc. | St-Rémi | QC | Type II | Industrial |
| 20-Jun-16 | Schlumberger Canada Limited | Calgary | AB | Type II | Industrial |
| 20-Jun-16 | Standard General Inc. | Calgary | AB | Type II | Industrial |
| 20-Jun-16 | Standard General Inc. | Calgary | AB | Type II | Industrial |
| 21-Jun-16 | Aecon Construction and Materials Limited | Caledon | ON | Type II | Industrial |
| 21-Jun-16 | Canadian Nuclear Safety Commission | Ottawa | ON | Type II | Academic & research |
| 21-Jun-16 | Groupe ABS Inc. | St-Rémi | QC | Type II | Industrial |
| 21-Jun-16 | Groupe ABS Inc. | St-Rémi | QC | Type II | Industrial |
| 22-Jun-16 | Institut de recherches cliniques de Montréal | Montréal | QC | Type II | Academic & research |
| 22-Jun-16 | Institut de recherches cliniques de Montréal | Montréal | QC | Type II | Medical |
| 22-Jun-16 | Nine Energy Canada Inc. | Clairmont | AB | Type II | Industrial |
| 22-Jun-16 | Troxler Canada Inc. | Laval | QC | Type II | Commercial |
| 22-Jun-16 | Troxler Canada Inc. | Laval | QC | Type II | Commercial |
| 23-Jun-16 | Bakos (N.D.T.) Inspection (1989) Ltd. | Whitecourt | AB | Type II | Industrial |
| 23-Jun-16 | Magnum Perforating Services Inc | Drayton Valley | AB | Type II | Industrial |
| 23-Jun-16 | Nelson's Welding Inspection Ltd. | Drayton Valley | AB | Type II | Industrial |
| 23-Jun-16 | SNC-Lavalin GEM Québec Inc. | Montréal | QC | Type II | Industrial |
| 27-Jun-16 | Centre intégré universitaire de santé et de services sociaux | Montréal | QC | Type II | Medical |
| 27-Jun-16 | Centre intégré universitaire de santé et de services sociaux | Montréal | QC | Type II | Commercial |
| 28-Jun-16 | Aecon Construction and Materials Limited | Caledon | ON | Type II | Industrial |
| 28-Jun-16 | Amec Foster Wheeler Americas Limited / Amec Foster Wheeler A | Lloydminster | AB | Type II | Industrial |
| 29-Jun-16 | 3M Canada Company | London | ON | Type II | Industrial |
| 29-Jun-16 | Alberta Agriculture and Rural Development | Lethbridge | AB | Type II | Industrial |
| 29-Jun-16 | Alberta Agriculture and Rural Development | Lethbridge | AB | Type II | Industrial |
| 29-Jun-16 | Coco Paving (1990) Inc. | Tecumseh | ON | Type II | Industrial |
| 29-Jun-16 | General Dynamics Produits de defense et Systemes Tactiques – Canada Inc. | Le Gardeur | QC | Type II | Academic & research |
| 29-Jun-16 | GHD Consultants Ltd. | Montréal | QC | Type II | Industrial |
| 29-Jun-16 | Tuboscope Vetco Canada ULC | Nisku | AB | Type II | Industrial |
| 30-Jun-16 | Soil Engineers Ltd. | Toronto | ON | Type II | Industrial |
| Inspection date | Licensee name | City | Province | Inpection type | Sector |
|---|---|---|---|---|---|
| 4-Jul-16 | Hoban Equipment Ltd. | Vernon | BC | Type II | Industrial |
| 4-Jul-16 | Hôpital de Verdun | Verdun | QC | Type II | Medical |
| 4-Jul-16 | Hôpital de Verdun | Verdun | QC | Type II | Medical |
| 5-Jul-16 | Millar Western Forest Products Ltd. | Whitecourt | AB | Type II | Industrial |
| 4-Jul-16 | Mitsubishi Hitachi Power Systems Canada, Ltd. | Saskatoon | SK | Type II | Industrial |
| 4-Jul-16 | MNA Engineering Ltd. | Scarborough | ON | Type II | Industrial |
| 4-Jul-16 | NARL Refining Inc. | Come By Chance | NL | Type II | Industrial |
| 4-Jul-16 | Pro-Test Professional Testing & Inspection Co. Ltd. | Winnipeg | MB | Type II | Industrial |
| 4-Jul-16 | Pro-Test Professional Testing & Inspection Co. Ltd. | Winnipeg | MB | Type II | Industrial |
| 4-Jul-16 | Stantec Consulting Ltd. | Barrie | ON | Type II | Industrial |
| 5-Jul-16 | 1583023 Alberta Ltd. | St. Albert | AB | Type II | Industrial |
| 5-Jul-16 | Acuren Inc. | Edmonton | AB | Type II | Industrial |
| 5-Jul-16 | Acuren Inc. | Edmonton | AB | Type II | Industrial |
| 5-Jul-16 | Acuren Inc. | Edmonton | AB | Type II | Industrial |
| 5-Jul-16 | Bunge Canada Holdings I ULC | Oakville | ON | Type II | Industrial |
| 5-Jul-16 | Canada Border Services Agency | Ottawa | ON | Type II | Industrial |
| 5-Jul-16 | Clifton Associates Ltd. | Regina | SK | Type II | Medical |
| 5-Jul-16 | Englobe Corp. | Laval | QC | Type II | Industrial |
| 5-Jul-16 | Pengrowth Corporation | Calgary | AB | Type II | Industrial |
| 5-Jul-16 | Schlumberger Canada Limited | Calgary | AB | Type II | Industrial |
| 5-Jul-16 | SNC-Lavalin Industrial Atlantic Inc. | Paradise | NL | Type II | Industrial |
| 5-Jul-16 | Sunnybrook Health Sciences Centre | Toronto | ON | Type II | Medical |
| 6-Jul-16 | Amec Foster Wheeler Americas Limited / Amec Foster Wheeler A | Lloydminster | AB | Type II | Industrial |
| 6-Jul-16 | A-Tech N.D.T. Limited | Whitecourt | AB | Type II | Industrial |
| 6-Jul-16 | Browning Harvey Limited | St John's | NL | Type II | Industrial |
| 6-Jul-16 | Clunie Consulting Engineers Ltd. | Prince Albert | SK | Type II | Industrial |
| 6-Jul-16 | Coco Paving (1990) Inc. | Tecumseh | ON | Type II | Industrial |
| 6-Jul-16 | Enbridge Employee Services Canada Inc. | Edmonton | AB | Type II | Industrial |
| 6-Jul-16 | EXP Services Inc. | Fredricton | NB | Type II | Industrial |
| 6-Jul-16 | GHD Consultants Ltd. | Mississauga | ON | Type II | Academic & research |
| 6-Jul-16 | Honeywell Ltd. | Lachine | QC | Type II | Commercial |
| 6-Jul-16 | Imperial Oil Limited/ Compagnie Pétrolière Impériale Ltée | East St. Paul | MB | Type II | Industrial |
| 6-Jul-16 | Iogen Corporation | Ottawa | ON | Type II | Industrial |
| 6-Jul-16 | IonBeam Applications S.A. | Louvain-la-Neuve | Belgium | Type II | Commercial |
| 6-Jul-16 | K+S Potash Canada General Partnership | Saskatoon | SK | Type II | Industrial |
| 6-Jul-16 | Labo S.M. Inc. | Longeuil | QC | Type II | Industrial |
| 6-Jul-16 | Merivale Medical Imagining Inc. | Nepean | ON | Type II | Medical |
| 6-Jul-16 | Pro-Test Professional Testing & Inspection Co. Ltd. | Winnipeg | MB | Type II | Industrial |
| 6-Jul-16 | Reliance OFS Canada Ltd. | Blackfalds | AB | Type II | Industrial |
| 6-Jul-16 | Suncor Energy Inc. | St. John's | NL | Type II | Industrial |
| 6-Jul-16 | The Eastern Regional Integrated Health Authority | St. John's | NL | Type II | Medical |
| 6-Jul-16 | The Eastern Regional Integrated Health Authority | St. John's | NL | Type II | Medical |
| 7-Jul-16 | Acuren Inc. | Edmonton | AB | Type II | Industrial |
| 7-Jul-16 | Acuren Inc. | Edmonton | AB | Type II | Industrial |
| 7-Jul-16 | Aecom Canada Ltd. | Edmonton | AB | Type II | Industrial |
| 7-Jul-16 | Amec Foster Wheeler Americas Limited / Amec Foster Wheeler A | Lloydminster | AB | Type II | Industrial |
| 7-Jul-16 | Englobe Corp. | Laval | QC | Type II | Industrial |
| 7-Jul-16 | Eng-Tech Consulting Limited | Winnipeg | MB | Type II | Industrial |
| 7-Jul-16 | Golder Associates Ltd. | Mississauga | ON | Type II | Industrial |
| 7-Jul-16 | Morey Associates Limited | Kemptville | ON | Type II | Industrial |
| 7-Jul-16 | Pembina Pipeline Corporation | Sherwood Park | AB | Type II | Industrial |
| 7-Jul-16 | SNC-Lavalin Inc. | Saskatoon | SK | Type II | Industrial |
| 7-Jul-16 | Stantec Consulting Ltd. | Barrie | ON | Type II | Industrial |
| 7-Jul-16 | The Eastern Regional Integrated Health Authority | St. John's | NL | Type II | Medical |
| 7-Jul-16 | The Eastern Regional Integrated Health Authority | St. John's | NL | Type II | Medical |
| 7-Jul-16 | TISI Canada Inc. | Oakville | ON | Type II | Industrial |
| 7-Jul-16 | Tusk Inspection Services Inc. | Fox Creek | AB | Type II | Industrial |
| 8-Jul-16 | Aker Solutions Asset Integrity and Management Canada Inc. | St. John's | NL | Type II | Industrial |
| 8-Jul-16 | Baker Hughes Canada Company | Leduc | AB | Type II | Industrial |
| 8-Jul-16 | EXP Services Inc. | Fredricton | MB | Type II | Industrial |
| 8-Jul-16 | Husky Oil Operations Limited | St. John's | NL | Type II | Industrial |
| 8-Jul-16 | Labatt Brewing Company Ltd. / La Brasserie Labatt limitée | London | ON | Type II | Industrial |
| 11-Jul-16 | Canadian Tower Scanning Inc. | Sarnia | ON | Type II | Industrial |
| 11-Jul-16 | Clifton Associates Ltd. | Regina | SK | Type II | Industrial |
| 11-Jul-16 | Stantec Consulting Ltd. | Barrie | ON | Type II | Industrial |
| 11-Jul-16 | WSP Canada | Red Deer | AB | Type I | Industrial |
| 12-Jul-16 | Canadian Natural Resources Limited | Bonnyville | AB | Type II | Industrial |
| 12-Jul-16 | Canadian Natural Resources Limited | Bonnyville | AB | Type II | Industrial |
| 12-Jul-16 | Cruickshank Construction Limited | Morrisburg | ON | Type II | Industrial |
| 12-Jul-16 | Cruickshank Construction Limited | Morrisburg | ON | Type II | Industrial |
| 12-Jul-16 | K.J. Beamish Construction Co., Limited | King City | ON | Type II | Industrial |
| 12-Jul-16 | Structural Inspections Limited | Milton | ON | Type II | Industrial |
| 12-Jan-16 | Terraprobe Testing Ltd. | Brampton | ON | Type II | Industrial |
| 12-Jul-16 | Terraprobe Testing Ltd. | Brampton | ON | Type II | Industrial |
| 13-Jul-16 | Clifton Associates Ltd. | Regina | SK | Type II | Industrial |
| 13-Jul-16 | Enbridge Employee Services Canada Inc. | Edmonton | AB | Type II | Industrial |
| 13-Jul-16 | Enbridge Employee Services Canada Inc. | Edmonton | AB | Type II | Industrial |
| 13-Jul-16 | Express Pipeline Ltd. | Sherwood Park | AB | Type II | Industrial |
| 13-Jul-16 | Q Test Inspection Ltd. | Sylvan Lake | AB | Type II | Industrial |
| 13-Jul-16 | Soil Engineers Ltd. | Toronto | ON | Type II | Industrial |
| 14-Jul-16 | Hunt Inspection Ltd. | Stettler | AB | Type II | Industrial |
| 14-Jul-16 | Insight Medical Holdings Ltd. | Edmonton | AB | Type II | Medical |
| 14-Jul-16 | Insight Medical Holdings Ltd. | Edmonton | AB | Type I | Medical |
| 14-Jul-16 | McClymont and Rak Engineers Inc. | Vaughan | ON | Type II | Industrial |
| 14-Jul-16 | Misericordia Community Hospital | Edmonton | AB | Type II | Medical |
| 14-Jul-16 | Misericordia Community Hospital | Edmonton | AB | Type II | Medical |
| 14-Jul-16 | Shad & Associates Inc. | Vaughan | ON | Type II | Industrial |
| 14-Jul-16 | TISI Canada Inc. | Oakville | ON | Type II | Industrial |
| 14-Jul-16 | Tuboscope Vetco Canada ULC | Nisku | AB | Type II | Industrial |
| 15-Jul-16 | The Cobalt Refinery Company Inc. | Fort Saskatchean | AB | Type II | Industrial |
| 16-Jul-16 | Sunnybrook Health Sciences Centre | Toronto | ON | Type II | Medical |
| 16-Jul-16 | Sunnybrook Health Sciences Centre | Toronto | ON | Type II | Medical |
| 18-Jul-16 | AM Inspection Limited | Cabri | SK | Type I | Industrial |
| 18-Jul-16 | Groupe ABS Inc. | St-Rémi | QC | Type II | Industrial |
| 18-Jul-16 | Labo S.M. Inc. | Longeuil | QC | Type II | Industrial |
| 18-Jul-16 | TechSpec NDT Limited | Grande Prairie | AB | Type II | Industrial |
| 18-Jul-16 | Ville de Montréal / Division de l'expertise et du soutien | Montréal | QC | Type II | Industrial |
| 19-Jul-16 | Alston Associates Inc. | Toronto | ON | Type II | Industrial |
| 19-Jul-16 | Canyon Technical Services Ltd. | Red Deer | AB | Type II | Medical |
| 19-Jul-16 | EXP Services Inc. | Brampton | ON | Type II | Industrial |
| 19-Jul-16 | Mistras Canada, Inc. | Olds | AB | Type II | Industrial |
| 19-Jul-16 | Stuart Hunt & Associates Ltd. | St. Albert | AB | Type II | Commercial |
| 19-Jul-16 | Superior General Partner Inc. | Toronto | ON | Type II | Industrial |
| 19-Jul-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| 20-Jul-16 | 20/20 ND Technology Inc. | Grande Prairie | AB | Type II | Industrial |
| 20-Jul-16 | Halliburton Canada | Edmonton | AB | Type II | Industrial |
| 20-Jul-16 | Inspectrum Testing Inc. | Grande Prairie | AB | Type II | Industrial |
| 20-Jul-16 | Inspectrum Testing Inc. | Grande Prairie | AB | Type II | Industrial |
| 20-Jul-16 | Schlumberger Canada Ltd. | Calgary | AB | Type II | Industrial |
| 20-Jul-16 | Schlumberger Canada Ltd. | Calgary | AB | Type II | Industrial |
| 20-Jul-16 | Stantec Consulting Ltd. | Barrie | ON | Type II | Industrial |
| 20-Jul-16 | Stantec Consulting Ltd. | Barrie | ON | Type II | Industrial |
| 20-Jul-16 | Stantec Consulting Ltd. | Barrie | ON | Type II | Industrial |
| 21-Jul-16 | Buffalo Inspection Services (2005) Inc. | Edmonton | AB | Type II | Industrial |
| 21-Jul-16 | City of Calgary | Calgary | AB | Type II | Industrial |
| 21-Jul-16 | Nortech Advanced N.D.T. Ltd. | Sherwood Park | AB | Type II | Industrial |
| 21-Jul-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| 24-Jul-16 | Acuren Inc. | Edmonton | AB | Type II | Industrial |
| 25-Jul-16 | Gamma-Tech Inspection Ltd. | Calgary | AB | Type II | Industrial |
| 25-Jul-16 | GeoPacific Consultants Ltd. | Vancouver | BC | Type II | Industrial |
| 25-Jul-16 | GeoPacific Consultants Ltd. | Vancouver | BC | Type II | Industrial |
| 25-Jul-16 | Process Research ORTECH Inc. | Mississauga | ON | Type II | Academic & research |
| 25-Jul-16 | RTD Quality Services Inc. | Burlington | ON | Type II | Industrial |
| 25-Jul-16 | The Pepsi Bottling Group (Canada), ULC | Winnipeg | MB | Type II | Industrial |
| 25-Jul-16 | University of British Columbia | Vancouver | BC | Type II | Medical |
| 25-Jul-16 | WAV Inspection Ltd. | Brooks | AB | Type II | Industrial |
| 26-Jul-16 | Acuren Inc. | Edmonton | AB | Type II | Industrial |
| 26-Jul-16 | Acuren Inc. | Edmonton | AB | Type II | Industrial |
| 26-Jul-16 | Aecon Transportation West Ltd. | Calgary | AB | Type II | Industrial |
| 26-Jul-16 | All Test International Inc. | Brooks | AB | Type II | Industrial |
| 26-Jul-16 | All Test International Inc. | Brooks | AB | Type II | Industrial |
| 26-Jul-16 | Almadon Holdings Ltd. | Calgary | AB | Type II | Medical |
| 26-Jul-16 | Almor Testing Services Ltd. | Calgary | AB | Type II | Industrial |
| 26-Jul-16 | Almor Testing Services Ltd. | Calgary | AB | Type II | Induatrial |
| 26-Jul-16 | Almor Testing Services Ltd. | Calgary | AB | Type II | Industrial |
| 26-Jul-16 | Cascades Canada ULC | Kingsey Falls | QC | Type II | Industrial |
| 26-Jul-16 | Department of Medical Imaging | Comox | BC | Type II | Medical |
| 26-Jul-16 | Lewkowich Engineering Associates Ltd. | Nanaimo | BC | Type II | Industrial |
| 26-Jul-16 | McElhanney Consulting Services Ltd. | Courtenay | BC | Type II | Industrial |
| 26-Jul-16 | Natural Resources Canada | Ottawa | ON | Type II | Industrial |
| 26-Jul-16 | Northwest Engineers | Surrey | BC | Type II | Industrial |
| 26-Jul-16 | Parkland Geotechnical Consulting Ltd. | Red Deer | AB | Type II | Industrial |
| 26-Jul-16 | St. Joseph's General Hospital | Comox | BC | Type II | Medical |
| 26-Jul-16 | Sylvia Fedrouk Canadian Centre for Nuclear Innovation Inc. | Saskatoon | SK | Type II | Commercial |
| 26-Jul-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| 27-Jul-16 | Acuren Inc. | Edmonton | AB | Type II | Industrial |
| 27-Jul-16 | Almor Testing Services Ltd. | Calgary | AB | Type II | Industrial |
| 27-Jul-16 | Davies Geotechnical Inc. | Delta | BC | Type II | Industrial |
| 27-Jul-16 | Industrial Radiography Supplies & Services Inc. | Edmonton | AB | Type II | Commercial |
| 27-Jul-16 | LAW Inspection Services Inc. | Lethbridge | AB | Type II | Industrial |
| 27-Jul-16 | LAW Inspection Services Inc. | Lethbridge | AB | Type II | Industrial |
| 27-Jul-16 | Mark Anthony Group Inc. | Delta | BC | Type II | Industrial |
| 5-Feb-16 | McElhanney Consulting Services Ltd. | Courtnenay | BC | Type II | Industrial |
| 27-Jul-16 | MNA Engineering Ltd. | Scarborough | ON | Type II | Industrial |
| 27-Jul-16 | MNA Engineering Ltd. | Scarborough | ON | Type II | Industrial |
| 27-Jul-16 | Peto MacCallum Ltd. | Kitchener | ON | Type II | Industrial |
| 27-Jul-16 | Peto MacCallum Ltd. | Kitchener | ON | Type II | Industrial |
| 27-Jul-16 | Terraprobe Testing Ltd. | Brampton | ON | Type II | Industrial |
| 27-Jul-16 | Tetra Tech EBA Inc. | Calgary | AB | Type II | Industrial |
| 27-Jul-16 | The Pepsi Bottling Group (Canada), ULC | Delta | BC | Type II | Industrial |
| 27-Jul-16 | Thurber Engineering Ltd. | Calgary | AB | Type II | Industrial |
| 28-Jul-16 | All Test International Inc. | Brooks | AB | Type II | Industrial |
| 28-Jul-16 | AR Geotechnical Engineering Ltd. | Medicine Hat | AB | Type II | Industrial |
| 28-Jul-16 | Catalyst Paper Corporation | Powell River | BC | Type II | Industrial |
| 28-Jul-16 | Constellation Brands Canada, Inc. | Niagara Falls | ON | Type II | Industrial |
| 28-Jul-16 | Graymar Equipment (2008) Inc. | New Westminster | BC | Type II | Industrial |
| 28-Jul-16 | Graymar Equipment (2008) Inc. | New Westminster | BC | Type II | Industrial |
| 28-Jul-16 | Graymar Equipment (2008) Inc. | New Westminster | BC | Type II | Industrial |
| 28-Jul-16 | Interlake Acquisition Corporation Limited | St. Catherines | ON | Type II | Industrial |
| 28-Jul-16 | Klohn Crippen Berger Ltd. | Vancouver | BC | Type II | Industrial |
| 28-Jul-16 | Lafarge Canada Inc. | Exshaw | AB | Type II | Industrial |
| 28-Jul-16 | LAW Inspection Services Inc. | Lethbridge | AB | Type II | Industrial |
| 28-Jul-16 | Molson Canada 2005 | Toronto | ON | Type II | Industrial |
| 28-Jul-16 | Saskatchewan Cancer Agency | Saskatoon | SK | Type II | Medical |
| 28-Jul-16 | Saskatchewan Cancer Agency | Saskatoon | SK | Type II | Medical |
| 28-Jul-16 | Weyerhaeuser Company Limited | Kenora | ON | Type II | Industrial |
| 29-Jul-16 | City of Vancouver | Vancouver | BC | Type II | Industrial |
| 29-Jul-16 | Isologic Innovative Radiopharmaceuticals Ltd. | Lachine | QC | Type II | Commercial |
| 29-Jul-16 | Isologic Innovative Radiopharmaceuticals Ltd. | Dorval | QC | Type II | Commercial |
| 29-Jul-16 | Reliance OFS Canada Ltd. | Blackfalds | AB | Type II | Industrial |
| 5-Aug-16 | Menu Foods Limited | Mississauga | ON | Type II | Industrial |
| 5-Aug-16 | St. Mary's Hospital Centre | Montréal | QC | Type II | Medical |
| 5-Aug-16 | St. Mary's Hospital Centre | Montréal | QC | Type II | Medical |
| 8-Aug-16 | Cott Corporation | Mississauga | ON | Type II | Industrial |
| 8-Aug-16 | Englobe Corp. | Laval | QC | Type II | Industrial |
| 8-Aug-16 | Mistras Canada, Inc. | Olds | AB | Type II | Industrial |
| 8-Jul-16 | Solmatech Inc. | Le Gardeur | QC | Type II | Industrial |
| 9-Aug-16 | Amec Foster Wheeler Americas Limited / Amec Foster Wheeler A | Lloydminster | AB | Type II | Industrial |
| 9-Aug-16 | Amec Foster Wheeler Americas Limited / Amec Foster Wheeler A | Lloydminster | AB | Type II | Industrial |
| 9-Aug-16 | Amec Foster Wheeler Americas Limited / Amec Foster Wheeler A | Lloydminster | BC | Type II | Industrial |
| 9-Aug-16 | Graham Bros. Construction Limited | Brampton | ON | Type II | Industrial |
| 9-Aug-16 | Graymar Equipment (2008) Inc. | New Westminster | BC | Type II | Industrial |
| 9-Aug-16 | Graymar Equipment (2008) Inc. | New Westminster | BC | Type II | Industrial |
| 10-Aug-16 | A & A Concrete X-Ray and Coring Ltd. | Langley | BC | Type II | Industrial |
| 10-Aug-16 | Acuren Inc. | Edmonton | AB | Type II | Industrial |
| 10-Aug-16 | Advance Testing Ltd. | Surrey | BC | Type II | Industrial |
| 10-Aug-16 | All Can Inspection Services (2011) Inc. | Edmonton | AB | Type II | Industrial |
| 10-Aug-16 | Thermo Gamma-Metrics LLC | San Diego | CA | Type II | Commercial |
| 11-Aug-16 | Advance Testing Ltd. | Surrey | BC | Type II | Industrial |
| 11-Aug-16 | Aecon Construction and Materials Limited | Caledon | ON | Type II | Industrial |
| 11-Aug-16 | Atlantic Steel Processing Inc. | Mississauga | ON | Type II | Industrial |
| 11-Aug-16 | City of Vancouver | Vancouver | BC | Type II | Industrial |
| 11-Aug-16 | City of Vancouver | Vancouver | BC | Type II | Industrial |
| 11-Aug-16 | Imperial Oil Resources Limited | East St. Paul | MB | Type II | Industrial |
| 11-Aug-16 | Imperial Oil Resources Limited | East St. Paul | MB | Type II | Industrial |
| 11-Aug-16 | Imperial Oil Resources Limited | East St. Paul | MB | Type II | Industrial |
| 11-Aug-16 | Imperial Oil Resources Limited | East St. Paul | MB | Type II | Industrial |
| 11-Aug-16 | KPGP Inc. | Gatineau | QC | Type II | Industrial |
| 11-Aug-16 | Le Groupe Dimension Multi Veterinaire Inc. | Lachine | QC | Type II | Medical |
| 11-Aug-16 | Le Groupe Dimension Multi Veterinaire Inc. | Lachine | QC | Type II | Medical |
| 11-Aug-16 | Metro Testing Laboratories Ltd. | Burnaby | BC | Type II | Industrial |
| 11-Aug-16 | Polar Pak Inc. | Brampton | ON | Type II | Industrial |
| 12-Aug-16 | Golder Associates Ltd. | Mississauga | ON | Type II | Industrial |
| 12-Aug-16 | Pembina Pipeline Corporation | Sherwood Park | AB | Type II | Industrial |
| 12-Aug-16 | Pembina Pipeline Corporation | Sherwood Park | AB | Type II | Industrial |
| 12-Aug-16 | Pembina Pipeline Corporation | Sherwood Park | AB | Type II | Industrial |
| 12-Aug-16 | Pembina Pipeline Corporation | Sherwood Park | AB | Type II | Industrial |
| 12-Aug-16 | Pembina Pipeline Corporation | Sherwood Park | AB | Type II | Industrial |
| 12-Aug-16 | Pembina Pipeline Corporation | Sherwood Park | AB | Type II | Industrial |
| 12-Aug-16 | Pembina Pipeline Corporation | Sherwood Park | AB | Type II | Industrial |
| 12-Aug-16 | Pembina Pipeline Corporation | Sherwood Park | AB | Type II | Industrial |
| 12-Aug-16 | Pembina Pipeline Corporation | Sherwood Park | AB | Type II | Industrial |
| 12-Aug-16 | Thurber Engineering Ltd. | Calgary | AB | Type II | Industrial |
| 12-Aug-16 | Thurber Engineering Ltd. | Calgary | AB | Type II | Industrial |
| 15-Aug-16 | De Beers Canada Inc. | Timmins | ON | Type II | Industrial |
| 15-Aug-16 | De Beers Victor Project Office | Timmins | ON | Type II | Industrial |
| 15-Aug-16 | EXL Engineering Inc. | Delta | BC | Type II | Industrial |
| 15-Aug-16 | TTES Consulting Inc. | MacGregor | MB | Type II | Industrial |
| 15-Aug-16 | University of British Columbia | Vancouver | BC | Type II | Academic & research |
| 15-Aug-16 | Vancouver Coastal Health Authority | Vancouver | BC | Type II | Medical |
| 15-Aug-16 | Vancouver Coastal Health Authority | Vancouver | BC | Type II | Medical |
| 15-Aug-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| 16-Augb-16 | 2273044 Ontario Inc. | Vaughan | ON | Type II | Medical |
| 16-Aug-16 | Acuren Group Inc. | Edmonton | AB | Type II | Industrial |
| 16-Aug-16 | Bunge Canada Holdings I ULC | Oakville | ON | Type II | Industrial |
| 16-Aug-16 | EXP Services Inc. | Brampton | QC | Type II | Industrial |
| 16-Aug-16 | Interior Health Authority | Vernon | BC | Type II | Medical |
| 16-Aug-16 | Interior Health Authority | Vernon | BC | Type II | Medical |
| 16-Aug-16 | Jim Dent Construction Ltd. | Hope | BC | Type II | Industrial |
| 16-Aug-16 | Kootenay Boundary Regional Hospital | Trail | BC | Type II | Medical |
| 16-Aug-16 | Kootenay Boundary Regional Hospital | Trail | BC | Type II | Medical |
| 16-Aug-16 | Louisiana – Pacific Canada Ltd. | Vancouver | BC | Type II | Industrial |
| 16-Aug-16 | PEI Cancer Treatment Centre | Charlottetown | PE | Type II | Medical |
| 16-Aug-16 | Sleeman Breweries Ltd. | Guelph | ON | Type II | Industrial |
| 16-Aug-16 | Timmins and District Hospital | Timmins | ON | Type II | Medical |
| 16-Aug-16 | Timmins and District Hospital | Timmins | ON | Type II | Medical |
| 16-Aug-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| 17-Aug-16 | C. Villeneuve Construction Co. Ltd. | Hearst | ON | Type II | Industrial |
| 17-Aug-16 | Detour Gold Corporation | Cochrane | ON | Type II | Industrial |
| 17-Aug-16 | Emil Anderson Construction Co. Ltd. | Hope | BC | Type II | Industrial |
| 17-Aug-16 | Interior Testing Services Ltd. | Kelowna | BC | Type II | Industrial |
| 17-Aug-16 | Labatt Brewing Company Ltd. / La Brasserie Labatt limitée | London | ON | Type II | Industrial |
| 17-Aug-16 | Manitoba Transportation and Government Services | West St. Paul | MB | Type II | Industrial |
| 17-Aug-16 | Novadaq Technologies Inc. | Burnaby | BC | Type II | Industrial |
| 17-Aug-16 | R.M. Belanger Limited | Chelmsford | ON | Type II | Industrial |
| 17-Aug-16 | R.M. Belanger Limited | Chelmsford | ON | Type II | Industrial |
| 17-Aug-16 | Slick Inspection Limited | Medicine Hat | AB | Type II | Industrial |
| 17-Aug-16 | Southlake Regional Health Centre | Newmarket | ON | Type II | Medical |
| 17-Augb-16 | Southlake Regional Health Centre | Newmarket | ON | Type II | Medical |
| 17-Augb-16 | Southlake Regional Health Centre | Newmarket | ON | Type II | Medical |
| 17-Aug-16 | Stasuk Testing & Inspection Ltd. | Burnaby | BC | Type II | Industrial |
| 17-Aug-16 | TBT Engineering Limited | Thunder Bay | ON | Type II | Industrial |
| 17-Aug-16 | Teck Metals Ltd. | Kimberly | BC | Type II | Industrial |
| 17-Aug-16 | Teranorth Construction & Engineering Ltd. | Sudbury | ON | Type II | Industrial |
| 17-Aug-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| 18-Aug-16 | Arthon Industries Limited | Kelowna | BC | Type II | Industrial |
| 18-Aug-16 | InterWrap Inc. | Vancouver | BC | Type II | Industrial |
| 18-Aug-16 | Manitoba Transportation and Government Services | West St. Paul | MB | Type II | Industrial |
| 18-Aug-16 | Manitoba Transportation and Government Services | West St. Paul | MB | Type II | Industrial |
| 18-Aug-16 | Primero Mining Corp. | Matheson | ON | Type II | Industrial |
| 18-Aug-16 | Teck Coal Limited | Sparwood | BC | Type II | Industrial |
| 18-Aug-16 | University of British Columbia | Vancouver | BC | Type II | Academic & research |
| 18-Aug-16 | University of British Columbia | Vancouver | BC | Type II | Academic & research |
| 18-Aug-16 | University of British Columbia | Vancouver | BC | Type II | Academic & research |
| 18-Aug-16 | University of British Columbia | Vancouver | BC | Type II | Academic & research |
| 18-Aug-16 | University of British Columbia | Vancouver | BC | Type II | Academic & research |
| 18-Aug-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| 19-Aug-16 | Kelowna General Hospital | Kelowna | BC | Type II | Medical |
| 19-Aug-16 | Kelowna General Hospital | Kelowna | BC | Type II | Medical |
| 19-Aug-16 | Kontzamanis, Graumann, Smith MacMillan Inc. | Winnipeg | MB | Type II | Industrial |
| 19-Aug-16 | The Corporation of the City of Timmins | Timmins | ON | Type II | Industrial |
| 19-Aug-16 | WSP Canada Inc | Red Deer | AB | Type II | Industrial |
| 22-Aug-16 | Golder Associates Ltd | Mississauga | ON | Type II | Industrial |
| 23-Aug-16 | Amhil Enterprises | Burlington | ON | Type II | Industrial |
| 23-Aug-16 | Amhil Enterprises | Burlington | ON | Type II | Industrial |
| 23-Aug-16 | Soil Engineers Ltd. | Toronto | ON | Type II | Industrial |
| 23-Aug-16 | Vale Canada Limited | Copper Cliff | ON | Type II | Industrial |
| 23-Aug-16 | Vale Canada Limited | Copper Cliff | ON | Type II | Industrial |
| 23-Aug-16 | Vale Canada Limited | Copper Cliff | ON | Type II | Industrial |
| 24-Aug-16 | Alston Associates Inc. | Toronto | ON | Type II | Industrial |
| 24-Aug-16 | Amec Foster Wheeler Americas Limited / Amec Foster Wheeler A | Lloydminster | AB | Type II | Industrial |
| 24-Aug-16 | Best Theratronics Ltd. | Ottawa | ON | Type II | Commercial |
| 24-Aug-16 | Coco Paving (1990) Inc. | Tecumseh | ON | Type II | Industrial |
| 24-Aug-16 | Golder Associates Ltd. | Mississauga | ON | Type II | Industrial |
| 24-Aug-16 | Isologic Innovative Radiopharmaceuticals Ltd. | Lacine | QC | Type II | Commercial |
| 24-Aug-16 | R.M. Belanger Limited | Chelmsford | ON | Type II | Industrial |
| 24-Aug-16 | Teranorth Construction & Engineering Limited | Sudbury | ON | Type II | Industrial |
| 24-Aug-16 | Terraprobe Testing Ltd. | Brampton | ON | Type II | Industrial |
| 25-Aug-16 | Glencore Canada Corporation | Onaping | ON | Type II | Industrial |
| 25-Aug-16 | Glencore Canada Corporation | Onaping | ON | Type II | Industrial |
| 25-Aug-16 | Golder Associates Ltd. | Mississauga | ON | Type II | Industrial |
| 25-Aug-16 | IKO Industries Ltd | Brampton | ON | Type II | Industrial |
| 25-Aug-16 | Interpaving Asphalt and Aggregate Supply Ltd. | Garson | ON | Type II | Industrial |
| 25-Aug-16 | Lavis Contracting Co. Limited | Clinton | ON | Type II | Industrial |
| 25-Aug-16 | The Regional Municipality of Halton | Oakville | ON | Type II | Industrial |
| 26-Aug-16 | Amec Foster Wheeler Americas Limited / Amec Foster Wheeler A | Lloydminster | AB | Type II | Industrial |
| 26-Aug-16 | Commandité Emballages Kruger Inc. | Montréal | QC | Type II | Industrial |
| 26-Aug-16 | GHD Consultants Ltd. | Montréal | QC | Type II | Industrial |
| 26-Aug-16 | Glencore Canada Corporation | Onaping | ON | Type II | Industrial |
| 26-Aug-16 | Honeywell Ltd. | Lachine | QC | Type II | Industrial |
| 29-Aug-16 | EXP Services Inc. | Brampton | ON | Type II | Industrial |
| 29-Aug-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| 29-Feb-16 | R-Metrics | Burlington | ON | Type II | Commercial |
| 30-Aug-16 | AGS Associates Inc. | Toronto | ON | Type II | Industrial |
| 30-Aug-16 | EXP Services Inc. | Brampton | ON | Type II | Industrial |
| 30-Aug-16 | GHD Consultants Ltd. | Montréal | QC | Type II | Industrial |
| 30-Aug-16 | KMH Cardiology Centres Incorporated | Mississauga | ON | Type II | Medical |
| 30-Aug-16 | Miller Group Inc. | North Bay | ON | Type II | Industrial |
| 30-Aug-16 | MNA Engineering Ltd. | Scarborough | ON | Type II | Industrial |
| 30-Aug-16 | MNA Engineering Ltd. | Scarborough | ON | Type II | Industrial |
| 30-Aug-16 | MNA Engineering Ltd. | Scarborough | ON | Type II | Industrial |
| 30-Aug-16 | North Bay General Hospital | North Bay | ON | Type II | Medical |
| 30-Aug-16 | North Bay General Hospital | North Bay | ON | Type II | Medical |
| 30-Aug-16 | St. Mary's General Hospital | Kitchener | ON | Type II | Medical |
| 30-Aug-16 | St. Mary's General Hospital | Kitchener | ON | Type II | Medical |
| 30-Aug-16 | St. Mary's General Hospital | Kitchener | ON | Type II | Medical |
| 30-Aug-16 | St. Mary's General Hospital | Kitchener | ON | Type II | Medical |
| 31-Aug-16 | Cambridge Memorial Hospital | Cambridge | ON | Type II | Medical |
| 31-Aug-16 | City of Calgary | Calgary | AB | Type II | Industrial |
| 31-Aug-16 | Huntsville District Memorial Hospital | Huntsville | ON | Type II | Medical |
| 31-Aug-16 | North York General Hospital | North York | ON | Type II | Medical |
| 31-Aug-16 | North York General Hospital | North York | ON | Type II | Medical |
| 31-Aug-16 | Terraprobe Testing Ltd. | Brampton | ON | Type II | Industrial |
| Inspection date | Licensee name | City | Province | Inpection type | Sector |
|---|---|---|---|---|---|
| 1-Sep-16 | Fowler Construction Company Ltd. | Bracebridge | ON | Type II | Industrial |
| 1-Sep-16 | KMH Cardiology Centres Incorporated | Mississauga | ON | Type II | Medical |
| 1-Sep-16 | KMH Cardiology Centres Incorporated | Mississauga | ON | Type II | Medical |
| 1-Sep-16 | Trillium Imaging Inc. | Toronto | ON | Type II | Medical |
| 1-Sep-16 | Tulloch Contract Administration Inc. | Thessalon | ON | Type II | Industrial |
| 2-Sep-16 | RTD Quality Services Inc. | Burlington | ON | Type II | Industrial |
| 2-Sep-16 | RTD Quality Services Inc. | Burlington | ON | Type II | Industrial |
| 6-Sep-16 | Alston Associates Inc. | Toronto | ON | Type II | Industrial |
| 6-Sep-16 | Apotex Inc. | Toronto | ON | Type II | Academic & research |
| 6-Sep-16 | Bare Contracting Services Ltd. | Mississauga | ON | Type II | Industrial |
| 6-Sep-16 | Beta Research Laboratories Ltd. | Calgary | AB | Type II | Industrial |
| 6-Sep-16 | Golder Associates Ltd. | Mississauga | ON | Type II | Industrial |
| 6-Sep-16 | Golder Associates Ltd. | Mississauga | ON | Type II | Industrial |
| 6-Sep-16 | Soil Engineers Ltd. | Toronto | ON | Type II | Industrial |
| 7-Sep-16 | ArcelorMittal Coteau-du-Lac Inc. | Coteau-du-Lac | QC | Type II | Industrial |
| 7-Sep-16 | ArcelorMittal Coteau-du-Lac Inc. | Coteau-du-Lac | QC | Type II | Industrial |
| 7-Sep-16 | Groupe ABS Inc. | St-Rémi | QC | Type II | Industrial |
| 7-Sep-16 | Sintra Inc. | Montréal | QC | Type II | Industrial |
| 7-Sep-16 | WorleyParsons Canada Services Ltd. | Calgary | AB | Type II | Industrial |
| 7-Sep-16 | WorleyParsons Canada Services Ltd. | Calgary | AB | Type II | Industrial |
| 8-Sep-16 | Englobe Corp. | Laval | QC | Type II | Industrial |
| 8-Sep-16 | EXP Services Inc. | Brampton | ON | Type II | Industrial |
| 8-Sep-16 | GHD Consultants Ltd. | Montréal | QC | Type II | Industrial |
| 8-Sep-16 | NOVA Chemicals Corporation | Calgary | AB | Type II | Industrial |
| 9-Sep-16 | Almadon Holdings Ltd. | Calgary | AB | Type II | Medical |
| 9-Sep-16 | Cordax Evaluation Tehnologies Inc. | Calgary | AB | Type II | Industrial |
| 9-Sep-16 | Ottawa Cardiovascular Centre – Orleans Inc. | Ottawa | ON | Type II | Medical |
| 9-Sep-16 | WSP Canada | Toronto | ON | Type II | Industrial |
| 9-Sep-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| 9-Sep-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| 12-Sep-16 | Breton N.D. Testing Incorporated | Reserve Mines | NS | Type II | Industrial |
| 12-Sep-16 | EXP Services Inc. | Brampton | ON | Type II | Industrial |
| 12-Sep-16 | Geowest Testing Services Ltd. | North Vancouver | BC | Type II | Industrial |
| 12-Sep-16 | Kelowna General Hospital | Kelowna | BC | Type II | Medical |
| 12-Sep-16 | Metro Testing Laboratories Ltd. | Burnaby | BC | Type II | Industrial |
| 12-Sep-16 | Nexen Inc. | Fort McMurray | AB | Type II | Industrial |
| 12-Sep-16 | Nexen Inc. | Fort McMurray | AB | Type II | Industrial |
| 12-Sep-16 | Pembina Pipeline Corporation | Sherwood Park | AB | Type II | Industrial |
| 13-Sep-16 | Amgen British Columbia Inc. | Burnaby | BC | Type II | Medical |
| 13-Sep-16 | Canada Border Services Agency | Ottawa | ON | Type II | Industrial |
| 13-Sep-16 | Cenovus FCCL Ltd. | Calgary | AB | Type II | Industrial |
| 13-Sep-16 | Corporation Cott | Pointe-Claire | QC | Type II | Industrial |
| 13-Sep-16 | Dominion Diamond Ekati Corporation | Yellowknife | NT | Type II | Industrial |
| 13-Sep-16 | GeoPacific Consultants Ltd. | Vancouver | BC | Type II | Industrial |
| 13-Sep-16 | Royal Inland Hospital | Kamloops | BC | Type II | Medical |
| 13-Sep-16 | Royal Inland Hospital | Kamloops | BC | Type II | Medical |
| 13-Sep-16 | St. Joseph's Health Centre | Toronto | ON | Type II | Medical |
| 13-Sep-16 | St. Joseph's Health Centre | Toronto | ON | Type II | Medical |
| 13-Sep-16 | Stuart Hunt & Associates Ltd. | St. Albert | AB | Type II | Commercial |
| 13-Sep-16 | Tetra Tech EBA Inc. | Calgary | AB | Type II | Industrial |
| 13-Sep-16 | UTC Fire & Security Canada Inc. operating as Chubb Edwards | Mississauga | ON | Type II | Commercial |
| 14-Sep-16 | Advance Testing Ltd. | Surrey | BC | Type II | Industrial |
| 14-Sep-16 | Chung & Vander Doelen Engineering Ltd. | Kitchener | ON | Type II | Industrial |
| 14-Sep-16 | Devon Canada Corporation | Calgary | AB | Type II | Industrial |
| 14-Sep-16 | Devon Canada Corporation | Calgary | AB | Type II | Industrial |
| 14-Sep-16 | Devon Canada Corporation | Calgary | AB | Type II | Industrial |
| 14-Sep-16 | Golder Associates Ltd. | Mississauga | ON | Type II | Industrial |
| 14-Sep-16 | Health Canada / Santé Canada | Ottawa | ON | Type II | Academic & research |
| 14-Sep-16 | Honeywell Ltd. | Lachine | QC | Type II | Commercial |
| 14-Sep-16 | Les entreprises Rolland inc. | St-Jérôme | QC | Type II | Industrial |
| 14-Sep-16 | New Gold Inc. | Kamloops | BC | Type II | Industrial |
| 14-Sep-16 | New Gold Inc. | Kamloops | BC | Type II | Industrial |
| 14-Sep-16 | Peto MacCallum Ltd. | Kitchener | ON | Type II | Industrial |
| 14-Sep-16 | Stantec Consulting Ltd. | Barrie | ON | Type II | Industrial |
| 14-Sep-16 | Tetra Tech EBA Inc. | Calgary | AB | Type II | Industrial |
| 15-Sep-16 | 4338626 Canada Inc. | Montréal | QC | Type II | Industrial |
| 15-Sep-16 | British Columbia Cancer Agency | Vancouver | BC | Type II | Medical |
| 15-Sep-16 | British Columbia Cancer Agency | Vancouver | BC | Type II | Medical |
| 15-Sep-16 | British Columbia Cancer Agency | Vancouver | BC | Type II | Medical |
| 15-Sep-16 | Cave Inspection Ltd. | Kitscoty | AB | Type II | Industrial |
| 15-Sep-16 | City of Vancouver | Vancouver | BC | Type II | Industrial |
| 15-Sep-16 | Dawson Construction Limited | Kamloops | BC | Type II | Industrial |
| 15-Sep-16 | Edge Wireline Inc. | Red Deer | AB | Type II | Industrial |
| 15-Sep-16 | JTI-Macdonald Corp | Montréal | ON | Type II | Industrial |
| 15-Sep-16 | Logco Wireline Services Ltd. | Calmar | AB | Type II | Industrial |
| 15-Sep-16 | Mistras Canada, Inc. | Olds | AB | Type II | Industrial |
| 15-Sep-16 | Steed and Evans Limited | St. Jacobs | ON | Type II | Industrial |
| 15-Sep-16 | Stuart Hunt & Associates Ltd. | St. Albert | AB | Type II | Commercial |
| 15-Sep-16 | Terracon Geotechnique Ltd. | Foer McMurray | AB | Type II | Industrial |
| 15-Sep-16 | Terracon Geotechnique Ltd. | Fort McMurray | AB | Type II | Industrial |
| 16-Sep-16 | Cave Inspection Ltd. | Kitscoty | AB | Type II | Industrial |
| 16-Sep-16 | Collège Ahuntsic | Montréal | QC | Type II | Academic & research |
| 16-Sep-16 | Collège Ahuntsic | Montréal | QC | Type I | Academic & research |
| 16-Sep-16 | Collège Ahuntsic | Montréal | QC | Type II | Academic & research |
| 16-Sep-16 | Collège Ahuntsic | Montréal | QC | Type II | Academic & research |
| 16-Sep-16 | Golder Associates Ltd. | Mississauga | ON | Type II | Industrial |
| 16-Sep-16 | Schlumberger Canada Limited | Calgary | AB | Type II | Industrial |
| 16-Sep-16 | Schlumberger Canada Limited | Calgary | AB | Type II | Industrial |
| 16-Sep-16 | Triquest Nondestructive Testing Corp. | Calgary | AB | Type II | Industrial |
| 16-Sep-16 | Triquest Nondestructive Testing Corp. | Calgary | AB | Type II | Induatrial |
| 19-Sep-16 | Foothills Radiography | Edson | AB | Type II | Industrial |
| 19-Sep-16 | Nortech Advanced N.D.T. Ltd. | Sherwood Park | AB | Type II | Industrial |
| 19-Sep-16 | Nortech Advanced N.D.T. Ltd. | Sherwood Park | AB | Type I | Industrial |
| 19-Sep-16 | Nortech Advanced N.D.T. Ltd. | Sherwood Park | AB | Type II | Industrial |
| 19-Sep-16 | P. Machibroda Engineering Ltd. | Saskatoon | SK | Type II | Industrial |
| 19-Sep-16 | Thurber Engineering Ltd. | Calgary | AB | Type II | Industrial |
| 19-Sep-16 | Weatherford Canada Ltd. | Calgary | AB | Type II | Industrial |
| 19-Sep-16 | Weatherford Canada Ltd. | Calgary | AB | Type II | Industrial |
| 19-Sep-16 | Weatherford Canada Partnership | Calgary | AB | Type II | Industrial |
| 20-Sep-16 | Black Creek Well Service Inc. | Oil Springs | ON | Type II | Industrial |
| 20-Sep-16 | Bluewater Health | Sarnia | ON | Type II | Medical |
| 20-Sep-16 | Centre Hospitalier de l'Université de Montréal | Montréal | QC | Type II | Medical |
| 20-Sep-16 | Centre Hospitalier de l'Université de Montréal | Montréal | QC | Type II | Medical |
| 20-Sep-16 | Centre Hospitalier de l'Université de Montréal | Montréal | QC | Type II | Medical |
| 20-Sep-16 | Centre Hospitalier de l'Université de Montréal | Montréal | QC | Type II | Medical |
| 20-Sep-16 | City of Ottawa / Ville d'Ottawa | Ottawa | ON | Type II | Industrial |
| 20-Sep-16 | Englobe Corp. | Laval | QC | Type II | Industrial |
| 20-Sep-16 | Englobe Corp. | Laval | QC | Type II | Industrial |
| 20-Sep-16 | Foothills Radiography | Edson | AB | Type II | Industrial |
| 20-Sep-16 | Imperial Oil Limited/ Compagnie Pétrolière Impériale Ltée | Edmonton | AB | Type II | Industrial |
| 20-Sep-16 | Innotech Inspection Solutions Ltd. | Sherwood Park | AB | Type II | Industrial |
| 20-Sep-16 | P. Machibroda Engineering Ltd. | Saskatoon | SK | Type II | Industrial |
| 20-Sep-16 | Prairie Mines & Royalty ULC | Edson | AB | Type II | Industrial |
| 20-Sep-16 | Prairie Mines & Royalty ULC | Edson | AB | Type II | Industrial |
| 20-Sep-16 | Saskatchewan Ministry of Highways and Infrastructure | Saskatoon | SK | Type II | Industrial |
| 20-Sep-16 | Westcoast Energy Inc. | Fort St. John | BC | Type II | Industrial |
| 20-Sep-16 | Westcoast Energy Inc. | Fort St. John | BC | Type II | Industrial |
| 20-Sep-16 | Westcoast Energy Inc. | Calgary | AB | Type II | Industrial |
| 21-Sep-16 | Agrium Inc. | Fort Saskatchewan | AB | Type II | Industrial |
| 21-Sep-16 | All Can Inspection Services (2011) Inc. | Edmonton | AB | Type II | Industrial |
| 21-Sep-16 | Amec Foster Wheeler Americas Limited / Amec Foster Wheeler A | Lloydminster | AB | Type II | Industrial |
| 21-Sep-16 | Baker Hughes Canada Company | Leduc | AB | Type II | Industrial |
| 21-Sep-16 | Carp Road Animal Hospital | Stittsville | ON | Type II | Medical |
| 21-Sep-16 | Grande Cache Coal Corporation | Grande Cache | AB | Type II | Industrial |
| 21-Sep-16 | IRISNDT Corp. | Edmonton | AB | Type II | Industrial |
| 21-Sep-16 | IRISNDT Corp. | Edmonton | AB | Type II | Industrial |
| 21-Sep-16 | Medical Imaging Centres Inc. | Mississauga | ON | Type II | Medical |
| 21-Sep-16 | Milner Power Inc | Grande Cache | AB | Type II | Industrial |
| 21-Sep-16 | Nortech Advanced N.D.T. Ltd. | Sherwood Park | AB | Type II | Industrial |
| 21-Sep-16 | Parkland Geotechnical Consulting Ltd. | Red Deer | AB | Type II | Industrial |
| 21-Sep-16 | Potash Corporation of Saskatchewan Inc. | Saskatoon | SK | Type II | Industrial |
| 22-Sep-16 | Altasteel Ltd. | Edmonton | AB | Type II | Industrial |
| 22-Sep-16 | Baker Hughes Canada Company | Calgary | AB | Type II | Industrial |
| 22-Sep-16 | Cal Frac Well Services Ltd. | Calgary | AB | Type II | Industrial |
| 22-Sep-16 | Cal Frac Well Services Ltd. | Calgary | AB | Type II | Industrial |
| 22-Sep-16 | Cody Last | Dawson Creek | BC | Type II | Industrial |
| 22-Sep-16 | Enbridge Employee Services Canada Inc. | Edmonton | AB | Type II | Industrial |
| 22-Sep-16 | Enbridge Employee Services Canada Inc. | Edmonton | AB | Type II | Industrial |
| 22-Sep-16 | Enbridge Employee Services Canada Inc. | Edmonton | AB | Type II | Industrial |
| 22-Sep-16 | Enbridge Employee Services Canada Inc. | Edmonton | AB | Type II | Industrial |
| 22-Sep-16 | Nortech Advanced N.D.T. Ltd. | Sherwood Park | AB | Type II | Industrial |
| 22-Sep-16 | Potash Corporation of Saskatchewan Inc. | Saskatoon | SK | Type II | Industrial |
| 22-Sep-16 | Potash Corporation of Saskatchewan Inc. | Saskatoon | SK | Type II | Industrial |
| 22-Sep-16 | Trican Well Service Ltd | Calgary | AB | Type II | Industrial |
| 23-Sep-16 | Alara Consultants Inc. | Edmonton | AB | Type II | Commercial |
| 23-Sep-16 | Alara Consultants Inc. | Edmonton | AB | Type II | Commercial |
| 23-Sep-16 | Cancer Imaging Services | Edmonton | AB | Type II | Academic & research |
| 23-Sep-16 | Cancer Imaging Services | Edmonton | AB | Type II | Medical |
| 26-Sep-16 | Centre Hospitalier de l'Université de Montréal | Montréal | QC | Type II | Medical |
| 26-Sep-16 | Parkland Geotechnical Consulting Ltd. | Red Deer | AB | Type II | Industrial |
| 27-Sep-16 | Milestone Engineering Services Ltd. | Wabasca | AB | Type II | Industrial |
| 27-Sep-16 | Tuboscope Vetco Canada ULC | Nisku | AB | Type II | Industrial |
| 28-Sep-16 | Daishowa-Marubeni International Limited | Peace River | AB | Type II | Industrial |
| 28-Sep-16 | Thurber Engineering Ltd. | Calgary | AB | Type II | Industrial |
| 28-Sep-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| 28-Sep-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| 29-Sep-16 | IRISNDT Corp. | Edmonton | AB | Type II | Industrial |
| 29-Sep-16 | Thurber Engineering Ltd. | Calgary | AB | Type II | Industrial |
| 29-Sep-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| 3-Oct-16 | RTD Quality Services Inc. | Burlington | ON | Type II | Industrial |
| 3-Oct-16 | SGS Canada Inc. | Lakefield | ON | Type II | Industrial |
| 3-Oct-16 | TISI Canada Inc. | Oakville | ON | Type II | Industrial |
| 4-Oct-16 | Acuren Inc. | Edmonton | AB | Type II | Industrial |
| 4-Oct-16 | Acuren Inc. | Edmonton | AB | Type II | Industrial |
| 4-Oct-16 | All Test International Inc. | Brooks | AB | Type II | Industrial |
| 4-Oct-16 | AVC Clinics BC Ltd. | Victoria | BC | Type II | Medical |
| 4-Oct-16 | Cargill Limited | Clavet | SK | Type II | Industrial |
| 4-Oct-16 | Golder Associates Ltd. | Mississauga | ON | Type II | Industrial |
| 4-Oct-16 | Mosaic Potash Colonsay ULC | Colonsay | SK | Type II | Industrial |
| 4-Oct-16 | RTD Quality Services Inc. | Burlington | ON | Type II | Industrial |
| 4-Oct-16 | Samuel, Son & Co. Limited | Stoney Creek | ON | Type II | Industrial |
| 4-Oct-16 | Sunshine Oilsands Ltd. | Calgary | AB | Type II | Industrial |
| 4-Oct-16 | Vancouver Island Health Authority | Victoria | BC | Type II | Medical |
| 4-Oct-16 | Vancouver Island Health Authority | Victoria | BC | Type II | Medical |
| 4-Oct-16 | WAV Inspection Ltd. | Brooks | AB | Type II | Industrial |
| 5-Oct-16 | Acuren Inc. | Edmonton | AB | Type II | Industrial |
| 5-Oct-16 | Bailey Metal Processing Ltd. | Concord | ON | Type II | Industrial |
| 5-Oct-16 | Canadian Natural Resources Limited | Fort McMurray | AB | Type II | Industrial |
| 5-Oct-16 | Canadian Natural Resources Limited | Fort McMurray | AB | Type II | Industrial |
| 5-Oct-16 | Ceda General Partners Ltd. | Sherwood Park | AB | Type II | Industrial |
| 5-Oct-16 | Endeavour Inspection Ltd. | Fort McMurray | AB | Type II | Industrial |
| 5-Oct-16 | Environment Canada | Saskatoon | SK | Type II | Academic & research |
| 5-Oct-16 | TISI Canada Inc. | Oakville | ON | Type II | Industrial |
| 5-Oct-16 | TISI Canada Inc. | Oakville | ON | Type II | Industrial |
| 5-Oct-16 | Vancouver Island Health Authority | Victoria | BC | Type II | Medical |
| 6-Oct-16 | Acuren Inc. | Edmonton | AB | Type II | Industrial |
| 6-Oct-16 | AM Inspection Limited | Cabri | SK | Type II | Industrial |
| 6-Oct-16 | ArcelorMittal Canada Inc. | Hamilton | ON | Type II | Industrial |
| 6-Oct-16 | ArcelorMittal Canada Inc. | Hamilton | ON | Type II | Commercial |
| 6-Oct-16 | Buffalo Inspection Services (2005) Inc. | Edmonton | AB | Type II | Industrial |
| 6-Oct-16 | Buffalo Inspection Services (2005) Inc. | Edmonton | AB | Type II | Industrial |
| 6-Oct-16 | Cave Inspection Inc | Wainwright | AB | Type II | Industrial |
| 6-Oct-16 | Chemtrade Fort McMurray GP Inc. | Fort Saskatchewan | AB | Type II | Industrial |
| 6-Oct-16 | Metalcare Group Inc. | Fort McMurray | AB | Type II | Industrial |
| 6-Oct-16 | Metro Testing Laboratories Ltd. | Burnaby | BC | Type II | Industrial |
| 6-Oct-16 | SNC-Lavalin GEM Québec Inc. | Montréal | QC | Type II | Industrial |
| 6-Oct-16 | Sweetcroft Engineering Consultants Ltd. | Caraquet | NB | Type II | Industrial |
| 6-Oct-16 | Terracon Geotechnique Ltd. | Fort McMurray | AB | Type II | Industrial |
| 6-Oct-16 | Terracon Geotechnique Ltd. | Fort McMurray | AB | Type II | Industrial |
| 6-Oct-16 | Terracon Geotechnique Ltd. | Fort McMurray | AB | Type II | Industrial |
| 7-Oct-16 | 1424624 Ontario Inc. | Milton | ON | Type II | Medical |
| 7-Oct-16 | Acuren Inc. | Edmonton | AB | Type II | Industrial |
| 7-Oct-16 | Endeavour Inspection Ltd. | Fort McMurray | AB | Type II | Industrial |
| 7-Oct-16 | Pinter & Associates Ltd | Saskatoon | SK | Type II | Industrial |
| 7-Oct-16 | Pinter & Associates Ltd | Saskatoon | SK | Type II | Industrial |
| 7-Oct-16 | University of Alberta | Edmonton | AB | Type II | Commercial |
| 7-Oct-16 | University of Victoria | Victoria | BC | Type II | Academic & research |
| 7-Oct-16 | University of Victoria | Victoria | BC | Type II | Academic & research |
| 7-Oct-16 | University of Victoria | Victoria | BC | Type II | Academic & research |
| 12-Oct-16 | Superior Metal Processing | Cambridge | ON | Type II | Industrial |
| 14-Oct-16 | Voith Canada Inc. | Hawkesbury | ON | Type II | Industrial |
| 17-Oct-16 | Nine Energy Canada Inc. | Clairmont | AB | Type II | Industrial |
| 18-Oct-16 | Atomic Inspection Services Ltd. | Fort St. John | BC | Type II | Industrial |
| 18-Oct-16 | Glencore Canada Corporation | Onaping | ON | Type II | Industrial |
| 18-Oct-16 | Golder Associates Ltd. | Mississauga | ON | Type II | Industrial |
| 18-Oct-16 | Interpaving Asphalt and Aggregate Supply Ltd. | Garson | ON | Type II | Industrial |
| 18-Oct-16 | Mevex Corporation | Stittsville | ON | Type II | Commercial |
| 18-Oct-16 | Natural Resources Canada/ Ressources naturelles Canada | Ottawa | ON | Type II | Academic & research |
| 18-Oct-16 | Sabia, Inc. | San Diego | CA | Type II | Commercial |
| 19-Oct-16 | 860851 Alberta Ltd. | Edmonton | AB | Type II | Industrial |
| 19-Oct-16 | Aecom Canada Ltd. | Edmonton | AB | Type II | Industrial |
| 19-Oct-16 | Aecon Construction and Materials Limited | Caledon | ON | Type II | Industrial |
| 19-Oct-16 | Atomic NDT Ltd. | Edmonton | AB | Type II | Industrial |
| 19-Oct-16 | Buffalo Inspection Services (2005) Inc. | Edmonton | AB | Type II | Industrial |
| 19-Oct-16 | Les Mines Agnico-Eagle Ltée / Agnico-Eagle Mines Ltd. | Baker Lake | NU | Type II | Industrial |
| 19-Oct-16 | Owl Inspection Services Ltd. | Fort St. John | BC | Type II | Industrial |
| 19-Oct-16 | Shawcor Ltd./Shawcor Ltée | Nisku | AB | Type II | Industrial |
| 20-Oct-16 | Deka Inspection Services Ltd. | Fort St. John | BC | Type II | Industrial |
| 20-Oct-16 | EXP Services Inc. / Les Services EXP Inc. | Laval | QC | Type II | Industrial |
| 20-Oct-16 | GHD Consultants Ltd. | Montréal | QC | Type II | Industrial |
| 20-Oct-16 | Hartstone Inc. | Okotoks | AB | Type II | Industrial |
| 20-Oct-16 | Labo S.M. Inc. | Longeuil | QC | Type II | Industrial |
| 20-Oct-16 | MNA Engineering Ltd. | Scarborough | ON | Type II | Industrial |
| 20-Oct-16 | Spectrum NDT Ltd. | Calgary | AB | Type II | Industrial |
| 20-Oct-16 | Willow Creek Coal Ltd. | Tumbler Ridge | BC | Type II | Industrial |
| 21-Oct-16 | British Columbia Cancer Agency | Abbotsford | BC | Type I | Medical |
| 21-Oct-16 | British Columbia Cancer Agency | Abbotsford | BC | Type II | Medical |
| 21-Oct-16 | British Columbia Cancer Agency | Abbotford | BC | Type II | Medical |
| 22-Oct-16 | Advance Testing Ltd. | Surrey | BC | Type II | Industrial |
| 24-Oct-16 | Mosaic Canada ULC | Regina | SK | Type II | Industrial |
| 24-Oct-16 | Mosaic Canada ULC | Regina | SK | Type II | Industrial |
| 24-Oct-16 | Mosaic Canada ULC | Regina | SK | Type II | Industrial |
| 24-Oct-16 | The University Hospital of Northern British Columbia | Prince George | BC | Type II | Medical |
| 24-Oct-16 | Yara Belle Plaine Inc. | Belle Plaine | SK | Type II | Industrial |
| 25-Oct-16 | Acuren Inc. | Edmonton | AB | Type II | Industrial |
| 25-Oct-16 | Canfor Pulp Ltd. | Prince George | BC | Type II | Industrial |
| 25-Oct-16 | Institut national de la recherche scientifique | Laval | QC | Type II | Academic & research |
| 25-Oct-16 | Institut national de la recherche scientifique | Laval | QC | Type II | Academic & research |
| 25-Oct-16 | Institut national de la recherche scientifique | Laval | QC | Type II | Medical |
| 25-Oct-16 | Regina Qu'Appelle Health Region | Regina | SK | Type II | Medical |
| 25-Oct-16 | Regina Qu'Appelle Health Region | Regina | SK | Type II | Medical |
| 25-Oct-16 | Regina Qu'Appelle Health Region | Regina | SK | Type II | Medical |
| 25-Oct-16 | Regina Qu'Appelle Health Region | Regina | SK | Type II | Medical |
| 25-Oct-16 | University of Regina | Regina | SK | Type II | Academic & research |
| 25-Oct-16 | University of Regina | Regina | SK | Type II | Academic & research |
| 25-Oct-16 | University of Regina | Regina | SK | Type II | Academic & research |
| 25-Oct-16 | Vancouver Coastal Health Authority | Vancouver | BC | Type II | Medical |
| 25-Oct-16 | Vancouver Coastal Health Authority | Vancouver | BC | Type II | Medical |
| 25-Oct-16 | Vancouver Coastal Health Authority | Vancouver | BC | Type II | Medical |
| 25-Oct-16 | Vancouver Coastal Health Authority | Vancouver | BC | Type II | Medical |
| 25-Oct-16 | Vancouver Coastal Health Authority | Vancouver | BC | Type II | Medical |
| 25-Oct-16 | Vancouver Coastal Health Authority | Vancouver | BC | Type II | Medical |
| 26-Oct-16 | Asphalte, Béton, Carrières Rive-Nord Inc. | Mirabel | QC | Type II | Industrial |
| 26-Oct-16 | Cariboo Pulp & Paper Company | Quesnel | BC | Type II | Industrial |
| 26-Oct-16 | Carleton University | Ottawa | ON | Type II | Academic & research |
| 26-Oct-16 | Carleton University | Ottawa | ON | Type II | Academic & research |
| 26-Oct-16 | Carleton University | Ottawa | ON | Type II | Academic & research |
| 26-Oct-16 | Carleton University | Ottawa | ON | Type II | Academic & research |
| 26-Oct-16 | Carleton University | Ottawa | ON | Type II | Academic & research |
| 26-Oct-16 | Carleton University | Ottawa | ON | Type II | Academic & research |
| 26-Oct-16 | Carleton University | Ottawa | ON | Type II | Academic & research |
| 26-Oct-16 | Cascades Canada ULC | Kingsey Falla | QC | Type II | Industrial |
| 26-Oct-16 | Clifton Associates Ltd. | Regina | SK | Type II | Industrial |
| 26-Oct-16 | Coco Paving (1990) Inc. | Tecumseh | ON | Type II | Industrial |
| 26-Oct-16 | Consumers' Co-Operative Refineries Ltd. | Regina | SK | Type II | Industrial |
| 26-Oct-16 | Consumers' Co-Operative Refineries Ltd. | Regina | SK | Type II | Industrial |
| 26-Oct-16 | Groupe ABS Inc. | St-Rémi | QC | Type II | Industrial |
| 26-Oct-16 | Honeywell Ltd. | Lachine | QC | Type II | Commercial |
| 26-Oct-16 | Hôpital Shriners pour l'Enfant/ Shriners Hospital for Children | Montréal | QC | Type II | Academic & research |
| 26-Oct-16 | Horizon Engineering Inc. | North Vancouver | BC | Type II | Industrial |
| 26-Oct-16 | Horizon Engineering Inc. | North Vancouver | BC | Type II | Industrial |
| 26-Oct-16 | Husky Oil Operations Limited | Prince George | BC | Type II | Industrial |
| 26-Oct-16 | Metro Testing Laboratories Ltd. | Burnaby | BC | Type II | Industrial |
| 26-Oct-16 | Quesnel River Pulp Company | Quesnel | BC | Type II | Industrial |
| 26-Oct-16 | Samuel, Son & Co. Limited | Stoney Creek | ON | Type II | Industrial |
| 26-Oct-16 | Samuel, Son & Co. Limited | Stoney Creek | ON | Type II | Industrial |
| 26-Oct-16 | TISI Canada Inc. | Oakville | ON | Type II | Industrial |
| 27-Oct-16 | Allnorth Consultants Limited | Terrace | BC | Type II | Industrial |
| 27-Oct-16 | AM Inspection Limited | Cabri | SK | Type II | Industrial |
| 27-Oct-16 | AR Geotechnical Engineering Ltd. | Medicine Hat | AB | Type II | Industrial |
| 27-Oct-16 | Centre intégré de santé et de services sociaux de la Montérégie-Est | St-Hyacinthe | QC | Type II | Medical |
| 27-Oct-16 | Centre intégré de santé et de services sociaux de la Montérégie-Est | St-Hyacinthe | QC | Type II | Medical |
| 27-Oct-16 | DWB Consulting Services Ltd. | Prince George | BC | Type II | Industrial |
| 27-Oct-16 | Metro Testing Laboratories Ltd. | Burnaby | BC | Type II | Industrial |
| 28-Oct-16 | Slick Inspection Limited | Medicine Hat | AB | Type II | Industrial |
| 30-Oct-16 | Solmatech Inc. | Le Gardeur | QC | Type II | Industrial |
| 31-Oct-16 | Aurora Inspection Limited | Sexsmith | AB | Type II | Industrial |
| 31-Oct-16 | Reliance OFS Canada Ltd. | Blackfalds | AB | Type II | Industrial |
| 31-Oct-16 | WSP Canada Inc. | Red Deer | AB | Type II | Industrial |
| Inspection date | Licensee name | City | Province | Inpection type | Sector |
|---|---|---|---|---|---|
| 1-Nov-16 | Atlas Testing Labs & Services (Nova Scotia) Ltd. | Salt Springs | NS | Type II | Industrial |
| 1-Nov-16 | City of Estevan | Estevan | SK | Type II | Industrial |
| 1-Nov-16 | City of Windsor | Windsor | ON | Type II | Industrial |
| 1-Nov-16 | Dalhousie University | Halifax | NS | Type II | Academic & research |
| 1-Nov-16 | Dalhousie University | Halifax | NS | Type II | Academic & research |
| 1-Nov-16 | EXP Services Inc. | Brampton | ON | Type II | Industrial |
| 1-Nov-16 | G.B. Contract Inspection Ltd. | Estevan | SK | Type II | Industrial |
| 1-Nov-16 | G.B. Contract Inspection Ltd. | Estevan | SK | Type II | Industrial |
| 1-Nov-16 | GHD Consultants Ltd. | Montréal | QC | Type II | Industrial |
| 1-Nov-16 | Oshanek Inspection Services (1972) Ltd. | Spruce Grove | AB | Type II | Industrial |
| 1-Nov-16 | Scapa Tapes North America Ltd. | Renfrew | ON | Type II | Industrial |
| 1-Nov-16 | SNC-Lavalin Inc. | Saskatoon | SK | Type II | Industrial |
| 1-Nov-16 | Tomahawk Inspection Inc. | Weyburn | SK | Type II | Industrial |
| 1-Nov-16 | Tomahawk Inspection Inc. | Weyburn | SK | Type II | Industrial |
| 2-Nov-16 | Acuren Inc. | Edmonton | AB | Type II | Industrial |
| 2-Nov-16 | Acuren Inc. | Regina | SK | Type II | Industrial |
| 2-Nov-16 | ADM Agri-Industries Company | Windsor | ON | Type II | Industrial |
| 2-Nov-16 | AM Inspection Limited | Cabri | SK | Type II | Industrial |
| 2-Nov-16 | AM Inspection Limited | Weyburn | SK | Type II | Industrial |
| 2-Nov-16 | Bonnett's Energy Services Ltd. | Grande Prairie | AB | Type II | Industrial |
| 2-Nov-16 | Brick Brewering Co. Limited | Kitchener | ON | Type II | Industrial |
| 2-Nov-16 | Cegep de Sainte-Foy | Québec | QC | Type II | Medical |
| 2-Nov-16 | Coco Paving (1990) Inc. | Tecumseh | ON | Type II | Industrial |
| 2-Nov-16 | DJ Galvanizing Corporation | Windsor | ON | Type II | Industrial |
| 2-Nov-16 | Michelin North America (Canada) Inc. | New Glasgow | NS | Type II | Industrial |
| 2-Nov-16 | Nova Scotia Power Incorporated | Sydney | NS | Type II | Industrial |
| 2-Nov-16 | Nova Scotia Power Incorporated | Trenton | NS | Type II | Industrial |
| 2-Nov-16 | Ray-Tech Inspection Inc. | Beaverlodge | AB | Type II | Industrial |
| 3-Nov-16 | ALSTOM Power Canada Inc. | New Waterford | NS | Type II | Industrial |
| 3-Nov-16 | AM Inspection Limited | Cabri | SK | Type II | Industrial |
| 3-Nov-16 | Brody Inspection Ltd. | Valleyview | AB | Type II | Industrial |
| 3-Nov-16 | Centre hospitalier de l'Université de Montréal | Montréal | QC | Type II | Medical |
| 3-Nov-16 | Coco Paving (1990) Inc. | Tecumseh | ON | Type II | Industrial |
| 3-Nov-16 | EXP Services Inc. | Fredricton | NB | Type II | Industrial |
| 3-Nov-16 | EXP Services Inc. | Fredricton | NB | Type II | Industrial |
| 3-Nov-16 | FB Nondestructive Examination Ltd. | Moose Jaw | SK | Type II | Industrial |
| 3-Nov-16 | FB Nondestructive Examination Ltd. | Moose Jaw | SK | Type II | Industrial |
| 3-Nov-16 | Golder Associates Ltd. | Mississauga | ON | Type II | Industrial |
| 3-Nov-16 | Knight Vision Inspections Inc. | Regina | SK | Type II | Industrial |
| 3-Nov-16 | Knight Vision Inspections Inc. | Regina | SK | Type II | Industrial |
| 3-Nov-16 | Riverview Steel Co. Ltd. | Windsor | ON | Type II | Industrial |
| 4-Nov-16 | S.G.H. Inspection Ltd. | Grovedale | AB | Type II | Industrial |
| 7-Nov-16 | Centre Intégré de Santé et de Services Sociaux de Laval | Laval | QC | Type II | Medical |
| 7-Nov-16 | Magna Exteriors Inc. | Guelph | ON | Type II | Industrial |
| 8-Nov-16 | Shell Lubricants | Brockville | ON | Type II | Industrial |
| 8-Nov-16 | University of Ottawa | Ottawa | ON | Type II | Academic & research |
| 14-Nov-16 | GHD Consultants Ltd. | Montréal | QC | Type II | Industrial |
| 14-Nov-16 | Les Pavages des Monts Inc. | Matane | QC | Type II | Industrial |
| 14-Nov-16 | University of Winnipeg | Winnipeg | MB | Type II | Academic & research |
| 14-Nov-16 | University of Winnipeg | Winnipeg | MB | Type II | Academic & research |
| 15-Nov-16 | College of the North Atlantic | Burin | NL | Type II | Industrial |
| 15-Nov-16 | College of the North Atlantic | Port aux Basques | NL | Type II | Industrial |
| 15-Nov-16 | Les Entreprises Mont Sterling Inc. | Sainte-Anne-des-Monts | QC | Type II | Industrial |
| 15-Nov-16 | Nuclear Management Co. Ltd. | Winnipeg | MB | Type II | Medical |
| 15-Nov-16 | Saskatchewan Power Corporation | Regina | SK | Type II | Industrial |
| 15-Nov-16 | Saskatchewan Power Corporation | Regina | SK | Type II | Industrial |
| 15-Nov-16 | Tembec Enterprises Inc./ Les Entreprises Tembec Inc. | Matane | QC | Type II | Industrial |
| 15-Nov-16 | University of Manitoba | Winnipeg | MB | Type II | Academic & research |
| 15-Nov-16 | Winpak Ltd. | Winnipeg | MB | Type II | Industrial |
| 16-Nov-16 | CancerCare Manitoba | Winnipeg | MB | Type II | Commercial |
| 16-Nov-16 | CancerCare Manitoba | Winnipeg | MB | Type II | Medical |
| 16-Nov-16 | CancerCare Manitoba | Winnipeg | MB | Type II | Comercial |
| 16-Nov-16 | Centre de santé et de services sociaux de la Côte-de-Gaspé | Gaspé | QC | Type II | Medical |
| 16-Nov-16 | Centre de santé et de services sociaux du Rocher-Percé | Chandler | QC | Type II | Medical |
| 16-Nov-16 | Centre intégré de santé et de services sociaux de la Gaspésie | Chandler | QC | Type II | Medical |
| 16-Nov-16 | Clifton Associates Ltd. | Regina | SK | Type II | Industrial |
| 16-Nov-16 | Construction DJL Inc. | Boucherville | QC | Type II | Industrial |
| 16-Nov-16 | Element Technical Services Inc. | Carlyle | SK | Type II | Industrial |
| 16-Nov-16 | Evraz Inc. NA Canada | Regina | SK | Type II | Industrial |
| 16-Nov-16 | Fisheries and Oceans Canada | Sydney | NS | Type II | Academic & research |
| 16-Nov-16 | Manitoba Infrastructure | West St. Paul | MB | Type II | Industrial |
| 16-Nov-16 | Tomahawk Inspection Inc. | Weyburn | SK | Type II | Industrial |
| 17-Nov-16 | Centre intégré de santé et de services sociaux de la Gaspésie | Maria | QC | Type II | Industrial |
| 17-Nov-16 | City of Winnipeg | Winnipeg | MB | Type II | Industrial |
| 17-Nov-16 | Construction DJL Inc. | Boucherville | QC | Type II | Industrial |
| 17-Nov-16 | Roke Technologies Ltd. | Calgary | AB | Type II | Industrial |
| 17-Nov-16 | Stantec Consulting Ltd. | Barrie | ON | Type II | Industrial |
| 17-Nov-16 | Tetra Tech EBA Inc. | Calgary | AB | Type II | Industrial |
| 18-Nov-16 | Clifton Associates Ltd. | Regina | SK | Type II | Industrial |
| 18-Nov-16 | Englobe Corp. | Laval | QC | Type II | Industrial |
| 18-Nov-16 | Groupe Lechasseur Ltée | Mont-Joli | QC | Type II | Industrial |
| 18-Nov-16 | Université de Montréal | Montréal | QC | Type I | Academic & research |
| 19-Nov-16 | Englobe Corp. | Laval | QC | Type II | Industrial |
| 22-Nov-16 | GeoPacific Consultants Ltd. | Vancouver | BC | Type II | Industrial |
| 22-Nov-16 | Schlumberger Canada Limited | Calgary | AB | Type II | Industrial |
| 23-Nov-16 | GeoPacific Consultants Ltd. | Vancouver | BC | Type II | Industrial |
| 23-Nov-16 | Landtek Limited | Hamilton | ON | Type II | Industrial |
| 23-Nov-16 | Quantum Petrophysics Inc. | Blackfalds | AB | Type II | Industrial |
| 23-Nov-16 | Trent University | Peterborough | ON | Type II | Academic & research |
| 24-Nov-16 | CGC Acquisition Corporation | Red Deer County | AB | Type II | Industrial |
| 24-Nov-16 | Health Science North | Sudbury | ON | Type II | Medical |
| 24-Nov-16 | Health Science North | Sudbury | ON | Type II | Medical |
| 24-Nov-16 | Hopital Maisonneuve-Rosemont | Montréal | QC | Type II | Medical |
| 24-Nov-16 | Hopital Maisonneuve-Rosemont | Montréal | QC | Type II | Medical |
| 24-Nov-16 | Johns Manville Canada Inc. | Innisfail | AB | Type II | Industrial |
| 24-Nov-16 | MyHealth Partners Inc. | Toronto | ON | Type II | Medical |
| 28-Nov-16 | Goba Associates Ltd. | Oshawa | ON | Type II | Industrial |
| 28-Nov-16 | SNC-Lavalin GEM Québec Inc. | Montréal | QC | Type II | Industrial |
| 28-Nov-16 | SNC-Lavalin GEM Québec Inc. | Montréal | QC | Type II | Industrial |
| 4-Dec-16 | SNC-Lavalin GEM Québec Inc. | Montréal | QC | Type II | Industrial |
| 5-Dec-16 | Buffalo Inspection Services (2005) Inc. | Edmonton | AB | Type II | Industrial |
| 5-Dec-16 | Buffalo Inspection Services (2005) Inc. | Edmonton | AB | Type II | Industrial |
| 5-Dec-16 | Triquest Nondestructive Testing Corp. | Calgary | AB | Type II | Industrial |
| 5-Dec-16 | Triquest Nondestructive Testing Corp. | Calgary | AB | Type II | Industrial |
| 6-Dec-16 | Buffalo Inspection Services (2005) Inc. | Edmonton | AB | Type II | Industrial |
| 6-Dec-16 | Building Products of Canada Corp. | Edmonton | AB | Type II | Industrial |
| 6-Dec-16 | University of Alberta | Edmonton | ON | Type II | Academic & research |
| 6-Dec-16 | University of Alberta | Edmonton | AB | Type II | Academic & research |
| 7-Dec-16 | Alberta Health Services | Edmonton | AB | Type II | Medical |
| 7-Dec-16 | Alberta Health Services | Edmonton | AB | Type II | Medical |
| 7-Dec-16 | All Can Inspection Services (2011) Inc. | Edmonton | AB | Type II | Industrial |
| 7-Dec-16 | Ciment Québec Inc. | Saint-Basile | QC | Type II | Industrial |
| 7-Dec-16 | Hexion Canada Inc. | Edmonton | AB | Type II | Industrial |
| 7-Dec-16 | PANalytical | Saint-Basile | QC | Type II | Commercial |
| 8-Dec-16 | Englobe Corp. | Laval | QC | Type II | Industrial |
| 8-Dec-16 | Insight Medical Holdings Ltd. | Edmonton | AB | Type II | Medical |
| 8-Dec-16 | RadTag Technologies Inc. | Edmonton | AB | Type II | Medical |
| 8-Dec-16 | Rivest Technologies Incorporated | Edmonton | AB | Type II | Industrial |
| 8-Dec-16 | Trillium Health Partners | Mississauga | ON | Type II | Medical |
| 8-Dec-16 | Trillium Health Partners | Mississauga | ON | Type II | Medical |
| 9-Dec-16 | Almadon Holdings Ltd. | Calgary | AB | Type II | Medical |
| 12-Dec-16 | Bruce Power Inc. | Tiverton | ON | Type II | Industrial |
| 12-Dec-16 | Bruce Power Inc. | Tiverton | ON | Type II | Industrial |
| 12-Dec-16 | Cal Frac Well Services Ltd. | Calgary | AB | Type II | Industrial |
| 12-Dec-16 | Logco Wireline Services Ltd. | Calmar | AB | Type II | Industrial |
| 13-Dec-16 | Alco Gas & Oil Production Equipment Ltd. | Edmonton | AB | Type II | Industrial |
| 13-Dec-16 | Anode NDT Ltd. | Grande Prairie | AB | Type II | Industrial |
| 13-Dec-16 | Centerline Geomatics Ltd. | Fort McMurray | AB | Type II | Industrial |
| 13-Dec-16 | Centerline Geomatics Ltd. | Fort McMurray | AB | Type II | Industrial |
| 13-Dec-16 | Covenant Health | Edmonton | AB | Type II | Medical |
| 13-Dec-16 | Fort McMurray Inspection and Testing Incorporated | Fort McMurray | AB | Type II | Industrial |
| 13-Dec-16 | Fugro Canada Corp. | Calgary | AB | Type II | Industrial |
| 13-Dec-16 | Grey Nuns Community Health Centre | Edmonton | AB | Type II | Medical |
| 13-Dec-16 | Inspectrum Testing Inc. | Grande Prairie | AB | Type II | Industrial |
| 13-Dec-16 | Peter Kiewit Infrastructure Co. | Edmonton | AB | Type II | Industrial |
| 14-Dec-16 | 20/20 ND Technology Inc. | Grande Prairie | AB | Type II | Industrial |
| 14-Dec-16 | Ceda General Partners Ltd. | Sherwood Park | AB | Type II | Industrial |
| 14-Dec-16 | Centre Hospitalier de l'Université de Montréal | Montréal | QC | Type II | Medical |
| 14-Dec-16 | Centre Hospitalier de l'Université de Montréal | Montréal | QC | Type II | Medical |
| 14-Dec-16 | Centre Hospitalier de l'Université de Montréal | Montréal | QC | Type II | Medical |
| 14-Dec-16 | Centre Hospitalier de l'Université de Montréal | Montréal | QC | Type II | Medical |
| 14-Dec-16 | Duncan Geomatics & Consulting Ltd. | Fort McMurray | AB | Type II | Industrial |
| 14-Dec-16 | E2K Engineering Ltd. | Calgary | AB | Type II | Industrial |
| 14-Dec-16 | Gamma Spec NDT Ltd. | Grande Prairie | AB | Type II | Industrial |
| 14-Dec-16 | RTD Quality Services Inc. | Burlington | ON | Type II | Industrial |
| 14-Dec-16 | Ultratest N.D.T. Services (2010) Inc. | Edmonton | AB | Type II | Industrial |
| 14-Dec-16 | XE Inspection Inc. | Fort McMurray | AB | Type II | Industrial |
| 15-Dec-16 | Canadian Inspection Ltd. | Edmonton | AB | Type II | Industrial |
| 15-Dec-16 | GeoPro Consulting Limited | Ricmond Hill | ON | Type II | Industrial |
| 15-Dec-16 | Intrepid NDE Testing Corp. | Grande Prairie | AB | Type II | Industrial |
| 15-Dec-16 | S.G.H. Inspection Ltd. | Grovedale | AB | Type II | Industrial |
| 15-Dec-16 | Sola Engineering Inc. | Vaughan | ON | Type II | Industrial |
| 15-Dec-16 | Suncor Energy Inc. | Calgary | AB | Type II | Industrial |
| 5-Feb-16 | Suncor Energy Inc. | Calgary | AB | Type II | Industrial |
| 16-Dec-16 | Inspectrum Testing Inc. | Grande Prairie | AB | Type II | Industrial |
| 16-Dec-16 | Recon Petrotechnologies Ltd. | Edmonton | AB | Type II | Industrial |
| 20-Dec-16 | Centre intégré de santé et de services | Lévis | QC | Type II | Medical |
| 20-Dec-16 | Centre intégré de santé et de services | Lévis | QC | Type II | Medical |
| 20-Dec-16 | Ministère des Transports, de la Mobilité durable et de l'Éle | Québec | QC | Type II | Industrial |
| 20-Dec-16 | Vale Newfoundland & Labrador Limited | Happy Valley-Goose Bay | NL | Type II | Industrial |
| 21-Dec-16 | Centre intégré de santé et de services sociaux de Chaudière- | Thetford Mines | QC | Type II | Medical |
| 21-Dec-16 | Centre intégré de santé et de services sociaux de Chaudière- | Thetford Mines | QC | Type II | Medical |
| 22-Dec-16 | Alara Consultants Inc. | Edmonton | AB | Type II | Commercial |
| 22-Dec-16 | Uni-Vert Tech Inc. | Montréal | QC | Type II | Commercial |
| 29-Dec-16 | Ontario Power Generation Inc. | Whitby | ON | Type II | Industrial |
| 29-Dec-16 | Ontario Power Generation Inc. | Whitby | ON | Type II | Commercial |
Appendix F: Compliance rating levels
The following rating levels, as shown in table 10, reflect the transition in rating terminology used by the CNSC. While inspection reports may still use the previous rating levels, licensees that use nuclear substances and radiation devices can expect this transition to take place in time.
| Previous rating level | Description | New rating level | Description |
|---|---|---|---|
| A | Exceeds expectations | FS | Fully satisfactory |
| B | Meets expectations | SA | Satisfactory |
| C | Improvement is required | BE | Below expectations |
| D | This area is seriously compromised | ||
| E | Breakdown | UA | Unacceptable |
Fully satisfactory (FS)
Compliance with regulatory requirements is fully satisfactory. Compliance within the area exceeds requirements and CNSC expectations. Compliance is stable or improving, and any problems or issues that arise are promptly addressed.
Satisfactory (SA)
Compliance with regulatory requirements is satisfactory. Compliance within the area meets requirements and CNSC expectations. Any deviation is only minor, and any issues are considered to pose a low risk to the achievement of regulatory objectives and CNSC expectations. Appropriate improvements are planned.
Below expectations (BE)
Compliance with regulatory requirements falls below expectations. Compliance within the area deviates from requirements or CNSC expectations to the extent that there is a moderate risk of ultimate failure to comply. Improvements are required to address identified weaknesses. The licensee or applicant is taking appropriate corrective action.
Unacceptable (UA)
Compliance with regulatory requirements is unacceptable, and is seriously compromised. Compliance within the overall area is significantly below requirements or CNSC expectations, or there is evidence of overall non-compliance. Without corrective action, there is a high probability that the deficiencies will lead to unreasonable risk. Issues are not being addressed effectively, no appropriate corrective measures have been taken, and no alternative plan of action has been provided. Immediate action is required.
Appendix G: Grading inspections
For all inspections, CNSC inspectors evaluate a licensee’s performance against regulatory requirements found in the Nuclear Safety and Control Act, its regulations, and conditions included in the licensee’s licence. During an inspection, the inspector verifies compliance with specific regulatory requirements and assigns a grade (i.e., a compliance rating) based on his or her observations. (Please refer to appendix F for information on the compliance ratings for inspection.) Each requirement is ranked according to the relative risk of the particular regulatory requirement: high, medium or low. The requirements are linked to a particular safety and control area (SCA), and each SCA has different numbers of requirements. The scope of the inspections determines which of the requirements are to be inspected. Please refer to appendix B for the list of all SCAs.
For the majority of licensees, inspection results are determined as follows:
- Inspection evidence is entered into a licensing and compliance system which uses a complex algorithm to calculate an overall grade for each SCA based on the inspector’s grades.
- The SCA grade is based on the worst grade of the high-risk requirements. The SCA grade will be the lowest grade assigned to a high-risk requirement by an inspector, unless an unacceptable rating was assigned to a medium-risk requirement. In cases where a medium-risk risk requirement has been assessed as unacceptable, then the SCA grade will be one grade lower than the lowest grade assigned to high-risk requirement.
- If no high-risk requirements were inspected, then the SCA grade equals the worst grade from the medium-risk requirements.
- If no high-risk or medium-risk requirements were inspected, then no grade is assigned for that SCA. In other words, no SCA grade is assigned if the data comes from low-risk requirements only.
For inspections not recorded in the licensing and compliance system, inspectors review each compliance expectation and determine the overall rating of the SCA based on the magnitude of the non-compliances.
Figure 60 shows a blank inspection worksheet used by inspectors to conduct a compliance inspection. This worksheet is specific to the use of portable gauges. Figure 61 shows criteria that may be used in inspections of accelerators and Class II facilities.
| SCA | Fully satisfactory (FS) | Satisfactory (SA) | Below expectations (BE) | Unacceptable (UA) | Mitigating factor | Aggravating factor | Grade | Explanation/ justification |
|---|---|---|---|---|---|---|---|---|
| Radiation protection | Radiation doses are equal to or less than the norm for the sector. Contamination, if applicable, did not affect a worker. | Increased dose below reportable limits. Contamination that could affect a worker. | Exposure to a worker in excess of regulatory limits. An incident that would result in a licensee exceeding action level limits (see section 6 of the Radiation Protection Regulations). Limited contamination that could affect a few persons or limited area. | Exposures to multiple workers in excess of regulatory limits. Widespread contamination to several persons or within a place. | ||||
| Physical design | No significant weaknesses in any element of the facility design. | Reduced redundancy that is not likely to prevent a safety-related system from meeting its design intent. | Compromise to barriers where defence in depth would be considered reduced, however redundancy remains. Compromise to safety due to a situation that was not previously evaluated and is believed to be probable. | Compromise to barriers where defence in depth would be considered inadequate. Compromise to safety due to a situation that was not previously evaluated and is believed to be probable. | ||||
| Operating performance | No significant lapses in conduct of licensed activities in accordance with licensee procedures or processes. | Partial failure to conduct licensed activities in accordance with one licensee procedure or processes. | Failure to conduct licensed activities in accordance with one or more licensee procedures and processes. | Widespread systemic failure to ensure licensed activities conducted according to licensee procedures and processes. | ||||
| Fitness for service | No significant risk that systems or components will not remain effective or that equipment will not be able perform its intended function when called upon to do so. | Partial failure to ensure single system or components remain effective or equipment is able to perform its intended function when called upon to do so. | Failure to ensure single system or components remain effective or equipment is able to perform its intended function when called upon to do so. | Widespread systemic failure to ensure systems and components remain effective and equipment is able to perform its intended function when called upon to do so. | ||||
| Security | No significant weaknesses in security. | Weaknesses in access control or barrier. | Failure in one or more barriers designed to delay access to security category I or II sources. | Widespread systemic failure to adhere to security plan. | ||||
| Packaging and transport | No significant weaknesses in packaging and transport procedures and processes. | Failure in one of the licensee’s packaging and transport procedures and processes. | Failure in one or more elements of the licensee’s packaging and transport procedures and processes. | Widespread systemic failure to adhere to licensee’s packaging and transport procedures and processes. |
Appendix H: Abbreviations and glossary
- ALARA
- as low as reasonably achievable
- AMP
- administrative monetary penalty
- CNSC
- Canadian Nuclear Safety Commission
- COMP
- Canadian Organization of Medical Physicists
- CRPA
- Canadian Radiation Protection Assocaition
- EDO
- exposure device operator
- GBq
- gigabecquerel
- IAEA
- International Atomic Energy Agency
- INES
- International Nuclear and Radiological Event Scale
- MBq
- megabecquerel
- mSv
- millisievert
- NEW
- nuclear energy worker
- NSCA
- Nuclear Safety and Control Act
- RSO
- radiation safety officer
- SCA
- safety and control area
- Sv
- Sievert
- TBq
- Terabecquerel
Glossary
- cyclotron
- A particle accelerator that speeds up particles in a circular motion until they hit a target at the perimeter of the cyclotron. Some cyclotrons are used to produce medical isotopes.
- effective dose
- The sum of the products, expressed in sieverts, obtained by multiplying the equivalent dose of radiation received by, and committed to, each organ or tissue set out in column 1 of an item of Schedule 1 by the weighting factor set out in column 2 of that item. (Source: Radiation Protection Regulations)
- enforcement actions
- The set of activities associated to compel a licensee back into compliance and to deter further non-compliances with the Nuclear Safety and Control Act, its regulations, and licences, decisions and certificates issued by the CNSC.
- exposure device
- A radiation device that is designed for carrying out gamma radiography, and includes any accessory to the device such as a sealed source assembly, a drive mechanism, a sealed source assembly guide tube and an exposure head. (Sources: Nuclear Substances and Radiation Devices Regulations; Packaging and Transport of Nuclear Substances Regulations, 2015)
- five-year dosimetry period
- The period of five calendar years beginning on January 1 of the year following the year in which the Radiation Protection Regulations came into force, and every subsequent period of five calendar years.
- fixed nuclear gauge
- A radiation device that is attached to a structure and enables the nuclear substance it holds to be used for its radiation properties to measure process-related parameters (such as liquid flow or liquid level).
- medical linear accelerator
- An accelerator that produces a collimated beam of high-energy photons (i.e., X-rays) that are used to deliver controlled doses of radiation for therapeutic purposes.
- natural background radiation
- Radiation that is emitted from naturally occurring radioactive materials and cosmic rays.
- nuclear energy worker
- A person who is required, in the course of his or her business or occupation in connection with a nuclear substance or nuclear facility, to perform duties in such circumstances that there is a reasonable probability that he or she may receive a dose of radiation that is greater than the prescribed limit for the general public. (Source: Nuclear Safety and Control Act)
- nuclear medicine technologist
- A medical radiation technologist certified by the Canadian Association of Medical Radiation Technologists. The nuclear medicine technologist works in the field of nuclear medicine and performs various duties such as preparing and administering radiopharmaceuticals, taking images of different organs and bodily structures, using computers to process data and enhance images, analyzing biological specimens and working closely with all members of the healthcare team.
- one-year dosimetry period
- The period of one calendar year beginning on January 1 of the year following the year in which the Radiation Protection Regulation came into force, and every subsequent period of one calendar. (Source: Radiation Protection Regulations)
- portable nuclear gauge
- A portable radiation device used to measure density, level, thickness or moisture content.
- prescribed equipment
The equipment prescribed by section 20 of the General Nuclear Safety and Control Regulations.
Section 20 states that each of the following items is prescribed equipment for the purposes of the Nuclear Safety and Control Act:- a package, special form radioactive material, low dispersible radioactive material, fissile-excepted radioactive material, radioactive material that has a basic radionuclide value that is not listed in the IAEA Regulations and an instrument or article that has an alternative activity limit for an exempt consignment, as those terms are defined in subsection 1(1) of the Packaging and Transport of Nuclear Substances Regulations, 2015
- a radiation device and a sealed source, as defined in section 1 of the Nuclear Substances and Radiation Devices Regulations
- Class II prescribed equipment, as defined in section 1 of the Class II Nuclear Facilities and Prescribed Equipment Regulations
- equipment that is capable of being used in the design, production, operation or maintenance of a nuclear weapon or nuclear explosive device
All controlled nuclear equipment is prescribed equipment for the purposes of the Nuclear Safety and Control Act, with respect to the import and export of that equipment.
- radiation device
- A device that contains more than the exemption quantity of a nuclear substance and that enables the nuclear substance to be used for its radiation properties for various purposes such as industrial radiography, oil exploration, road construction and industrial processes.
- radiopharmaceutical
- A drug containing a radioactive substance that is used in medical imaging and cancer treatment.
- sealed source
- A radioactive nuclear substance in a sealed capsule or in a cover to which the substance is bonded, where the capsule or cover is strong enough to prevent contact with or the dispersion of the substance under the conditions for which the capsule or cover is designed. (Sources: Class I Nuclear Facilities Regulations; Class II Nuclear Facilities and Prescribed Equipment Regulations; Nuclear Substances and Radiation Devices Regulations)
- unsealed source
- A source other than a sealed source. (Source: Nuclear Substances and Radiation Devices Regulations) These nuclear substances are in a physical form where dispersion of the radioactive material is possible during use or handling. Usually a liquid, they may also be in solid, powder or gaseous form. Unsealed sources are commonly used in medical diagnostic and therapeutic treatments, as well as in laboratory research applications. Also called open source.
Appendix I: Relevant Regulatory References
The following are a list of regulatory references that apply to the use of nuclear substances and prescribed equipment. The list is not exhaustive.
Act and regulations
- Nuclear Safety and Control Act
- General Nuclear Safety and Control Regulations
- Administrative Monetary Penalties Regulations (Canadian Nuclear Safety Commission)
- Radiation Protection Regulations
- Class II Nuclear Facilities and Prescribed Equipment Regulations
- Nuclear Substances and Radiation Devices Regulations
- Packaging and Transport of Nuclear Substances Regulations, 2015
- Transportation of Dangerous Goods Regulations
Regulatory documents
- REGDOC-1.4.1, Licence Application Guide: Class II Nuclear Facilities and Prescribed Equipment (draft)
- REGDOC-1.5.1, Application Guide: Certification of Radiation Devices or Class II Prescribed Equipment (draft)
- REGDOC-1.6.1, Licence Application Guide: Nuclear Substances and Radiation Devices, Version 2
- REGDOC-2.2.3, Personnel Certification: Radiation Safety Officers
- REGDOC-2.2.3, Personnel Certification: Exposure Device Operators
- G-129, rev. 1, Keeping Radiation Exposures and Doses "As Low as Reasonably Achievable (ALARA)"
- REGDOC-2.9.1, Environmental Protection: Environmental Principles, Assessments and Protection Measures, Version 1.1
- REGDOC-2.12.3, Security of Nuclear Substances: Sealed Sources
- REGDOC-2.14.1, Information Incorporated by Reference in Canada’s Packaging and Transport of Nuclear Substances Regulations, 2015
Footnotes
- Footnote 1
-
Information about the categorization of sealed sources and their relative risks can be found on the CNSC website
- Footnote 2
-
Exceptions may be made for certain lower risk activities, such as the operation of mobile industrial accelerators and oil well logging accelerators.
- Footnote 3
-
All 14 orders were issued by inspectors. Five of the eight AMPs were issued following, or in conjunction with, orders.
Page details
- Date modified: